Imaging and data analysis is an essential step in the western blotting workflow. Western blots can be imaged and analyzed using different methods. This page reviews some of these methods such as luminescence detection, fluorescence imaging, densitometery, and autoradiography. It also reviews some of the analysis software products that Bio-Rad offers and their features.
Related Topics: Protein Blotting Methods, Protein Blotting Equipment, Membranes and Blotting Papers, Transfer Buffers, and Transfer Conditions.
Page Contents
Several instruments and devices are employed to document western blotting results.
- Densitometers — based on high-performance document scanners with minor modifications (leak-resistant scanning surface, built-in calibration tool); they utilize visible light for the analysis of electrophoresis gels (transmission mode) and blots (reflective mode) stained with visible dyes
- CCD (charge-couple device) cameras — versatile systems that can image gels and blots; they can operate with either transillumination provided by light boxes (visible or UV) positioned underneath the gel for imaging a variety of stains (Coomassie, silver, fluorescence) or epi-illumination of blots detected using colorimetric or fluorescence techniques. Different illumination wavelengths are available for multiplex fluorescence immunodetection. CCD cameras can also be used to detect chemiluminescent signals for which illumination is not needed. Super-cooled CCD cameras reduce image noise, allowing detection of faint luminescent signals with high sensitivity
- Laser-based imagers — offer the highest sensitivity, resolution, and linear dynamic range and are powerful image acquisition tools for gels and blots with proteins fluorescently labeled or stained with fluorescent dyes such as SYPRO Ruby or Flamingo. These imagers can be configured with lasers of different wavelengths, allowing single- or multiplex fluorescence detection. Laser scanners allow selective detection of multiple fluorescence labels on gels and blots
- Phosphor imagers — laser-based systems capable of imaging storage phosphor screens, which form a latent image when exposed to gamma and beta radiation. These screens have a large dynamic range and offer excellent sensitivity and quantitative accuracy with imaging times that are a fraction of those for film
- X-ray film — widely used for imaging autoradiographic and chemiluminescent blots but suffers from a limited dynamic range as well as a nonlinear response through this range. This method also requires the investment in and maintenance of a film developer and often requires processing multiple film sheets to obtain a usable image. Decreasing costs, superior quantitative data quality, and better ease of use are making CCD cameras and imagers preferred over film for these detection techniques
Comparison of western blot documentation and analysis methods.
| Imaging System | ||||||
|---|---|---|---|---|---|---|
| Gel Doc | ChemiDoc | |||||
| EZ | XR+ | XRS+ | Touch | MP | GS-900 | |
| Immunodetection | ||||||
| Chemiluminescence | ✓ | ✓ | ✓ | |||
| Colorimetric | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Fluorescence | ✓ | ✓ | ||||
| Autoradiography | ||||||
| Chemifluorescence | ✓ | |||||
| Total Protein Stain | ||||||
| Colorimetric | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Fluorescent | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Stain-free | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Excitation Type | ||||||
| Trans UV/Vis | ✓ | ✓ | ✓ | ✓ | ✓ | N/A |
| Epi white | ✓ | ✓ | ✓ | ✓ | N/A | |
| LED RGB | ✓ | N/A | ||||
| Laser RGB | N/A | |||||
| Imager Type | ||||||
| CCD | ✓ | ✓ | ||||
| Supercooled CCD | ✓ | ✓ | ✓ | |||
| Densitometer | ✓ | |||||
* Only 532 nm laser available.
For chemiluminescence detection, CCD imaging is the easiest, most accurate, and fastest method. Traditionally, the chemiluminescent signal from blots was detected by X-ray film. Film is a sensitive medium for capturing the chemiluminescent signal but suffers from a sigmoidal response to light with a narrow region of linear response, which limits its dynamic range. To gather information from a blot that has both intense and weak signals, multiple exposures are required to produce data for all samples in the linear range of the film. A process termed preflashing can improve linearity, but this requires extra equipment and effort. Additionally, quantitation of data collected by exposure to film requires digitization (that is, scanning of X-ray film with a densitometer). Bio-Rad's Clarity Western ECL Substrate is compatible with any HRP-conjugate secondary detection reagent and ideal for both digital and film-based imaging.
CCD cameras have a linear response over a broad dynamic range — 2–5 orders of magnitude — depending on the bit depth of the system. CCD cameras also offer convenience by providing a digital record of experiments for data analysis, sharing, and archiving, and by eliminating the need to continually purchase consumables for film development. The ChemiDoc Touch Imaging System offers chemiluminescent sensitivity equal to film.
Fluorescence, chemifluorescence, and colorimetric detection all benefit from the advantages of digital imaging: convenience, digital records of experiments, high sensitivity, and wide dynamic ranges. Fluorescent and chemifluorescent signals can be detected with different types of imaging systems, including CCD and laser-based technologies. For example, the ChemiDoc MP System can be used similarly to detect fluorescent and chemifluorescent signals. The decision to use one type of technology over another depends on budget and requirements for limit of detection and resolution. CCD systems are generally less expensive than laser-based systems. The resolution of CCD and laser-based systems can be similar, with the finest resolution settings of 50 µm when used for gels and blots. Another advantage of fluorescence and chemifluorescence detection is that the detection limits and dynamic range of CCD and laser-based systems generally far exceed the dynamic ranges of the fluorescence assays currently used for protein detection.
Colorimetric samples can be easily recorded and analyzed with a densitometer such as the GS-900 Calibrated Densitometer. The densitometer provides a highly reproducible digital record of the blot with excellent image resolution and accurate quantitation. The GS-900 Densitometer uses red, green, and blue color CCD technology to enhance detection of a wide variety of colorimetric staining reagents.
Autoradiography on X-ray film is the most widely used method to detect commonly used beta-emitting radioisotopes such as 35S, 32P, 33P, 12C, and 125I. Autoradiography provides a good combination of sensitivity and resolution without a large investment in detection substrates or imaging systems. For direct autoradiography the response of the film is linear only within a range of 1–2 orders of magnitude. Use of intensifying screens and fluorographic scintillators can increase sensitivity, and pre-exposing film to a flash of light can improve linearity. However, these measures to improve signal detection are limited. Phosphor imagers, such as Bio-Rad's PharosFX Plus System or Personal Molecular Imager™ (PMI) System, offer an alternative for detecting gels and blots labeled with beta-emitting radioisotopes. The initial investment in instrumentation frees up time and resources associated with using X-ray film and offers increased sensitivity, wider dynamic range, and 10 to 20 times shorter exposure times than those for X-ray film detection. The ability to accurately quantitate data is much greater with storage phosphor screens because the linear dynamic range of phosphor imagers is significantly greater – 5 orders of magnitude – enabling accurate quantitation and the elimination of overexposure and saturated signals.
Blot detection using an imaging system needs a robust software package for image acquisition. In addition, a good software package can magnify, rotate, resize, overlay, and annotate the corresponding gel and blot images, allowing export of the images to common documentation software. A good software package also allows analysis of the blot image and comparisons of relative signal intensities, protein molecular weights, and other data.
For automated acquisition and analysis of gel and blot images, Bio-Rad offers:
- Image Lab Software — control software for a variety of Bio-Rad imaging systems, Image Lab Software automatically determines the image with the best signal-to-noise ratio and generates a report. It also provides sophisticated algorithms to determine the number of lanes and bands in the image. The software can measure total and average quantities and determine relative and actual amounts of protein. This software is also capable of determining the presence/absence and up/down regulation of bands, their molecular weights, and other values. Signal intensities can be quantitated and compared to determine relative signal and to generate other data such as relative front values for molecular weight determinations
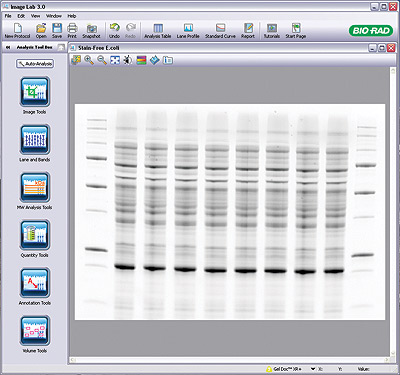
Image Lab Software. E. coli lysate was separated and activated on a 4–20% Criterion TGX Stain-Free Gel and transferred onto a PVDF membrane. The membrane was imaged on a Gel Doc EZ System and analyzed using Image Lab 3.0 Software.
- Quantity One 1-D Analysis Software — acquires, quantitates, and analyzes a variety of data, including radioactive, chemiluminescent, fluorescent, and color-stained samples acquired from densitometers, storage phosphor imagers, fluorescence imagers, and gel documentation systems. The software allows automatic configuration of these imaging systems with appropriate filters, lasers, LEDs, and other illumination sources and allows manual or automated analysis of SDS-PAGE gels and western blots
- PDQuest 2-D Analysis Software — used for efficient and accurate 2-D gel electrophoresis analysis
