For almost 50 years, immunoassays have allowed sensitive and highly specific detection of analytes of interest in biological samples for use in both life science research and clinical diagnostics. Traditional enzyme-linked immunosorbent assays (ELISA assays) detect and measure a single analyte per plate. Newer bead-based immunoassays, like those based on Luminex xMAP technology, utilize the same principle and apply it to uniquely identifiable beads. These beads allow for the simultaneous detection of multiple analytes in a single well or reaction. Multiplex immunoassays yield a wealth of information on the roles of multiple proteins and other biomolecules in diverse biological processes, thereby providing clinicians with insight into the identification and assessment of disease progression.
NEW PRODUCTBio-Plex SARS-CoV-2 Assays
Quickly assay SARS-CoV-2 variant neutralizing antibodies.
Page Contents
The first immunoassay was developed by Yalow and Berson (1960), who received the Nobel Prize for their efforts to measure insulin levels. These initial assays used radiolabels for detection. The radioimmunoassay (RIA) would remain the standard for the detection of bioanalytes for more than ten years because of its extraordinary sensitivity, despite the health risks and disposal issues posed by the use of radioisotopes. The search for a suitable alternative to the RIA led to the development of ELISA in the early 1970s (Engvall and Perlmann 1971, Van Weeman and Schuurs 1971). ELISA uses an enzymatic reaction as the basis of detection, rather than a radioactive signal. While early versions did not rival the sensitivity of the RIA, the development of highly specific monoclonal antibodies and chemiluminescence detection resulted in ELISA assays with sensitivity that exceeds that of radiolabels. Today, key advantages of ELISA are its ease of use, flexibility, and low cost. The impact of immunoassays on life science research and clinical diagnostics has been enormous, with almost 10,000 studies published per year that include the terms “enzyme immunoassay” and “enzyme-linked immunoassay” (Lequin 2005). The growth of proteomics and genomic analysis is driving the need to discover and monitor large numbers of biomarkers indicative of human disease states. The output of the Human Genome Project, for example, provides the ability to simultaneously monitor the roles of multiple genes during investigations of complex biological systems.
The Bio-Plex® multiplex immunoassay system utilizes xMAP technology licensed from Luminex to permit the multiplexing of up to 100 different assays within a single sample. This technique involves 100 distinctly colored bead sets created by the use of two fluorescent dyes at distinct ratios. These beads can be further conjugated with a reagent specific to a particular bioassay. The reagents may include antigens, antibodies, oligonucleotides, enzyme substrates, or receptors. The technology enables multiplex immunoassays in which one antibody to a specific analyte is attached to a set of beads with the same color, and the second antibody to the analyte is attached to a fluorescent reporter dye label. The use of different colored beads enables the simultaneous multiplex detection of many other analytes in the same sample. A dual detection flow cytometer is used to sort out the different assays by bead colors in one channel and determine the analyte concentration by measuring the reporter dye fluorescence in another channel (Figure 1).
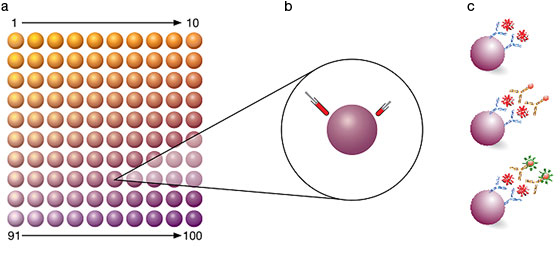
Fig 1. Multiplex immunoassay technology. Beads are colored internally with two different fluorescent dyes (red and infrared). Different concentrations of red and infrared dyes are used to generate up to 100 distinct bead regions. Each bead region is conjugated to a specific target analyte (a) followed by binding with a biotinylated detection antibody (b) and a reporter dye, streptavidin-conjugated phycoerythrin (c).
During data acquisition, the contents of each microplate well are drawn into the array reader, depending on the type of reader — either the flow cytometry/laser excitation–based Bio-Plex 200 and Bio-Plex 3D systems or the light emitting diode (LED)/image-based analysis employed in the Bio-Plex® MAGPIX™ multiplex reader. For the flow cytometry–based systems, precision fluidics align the beads in single file through a flow cell where two lasers excite the beads individually. The red classification laser excites the dyes in each bead, identifying its spectral address. The green reporter laser excites the reporter molecule associated with the bead, which allows quantitation of the captured analyte. With the LED/image-based Bio-Plex MAGPIX reader, the beads are drawn into a chamber and magnetically immobilized. Classification and reporter excitation is accomplished with the use of LEDs rather than lasers. In all readers, high-speed digital signal processors and software record the fluorescent signals simultaneously for each bead, translating the signals into data for each bead-based assay (Figure 2).
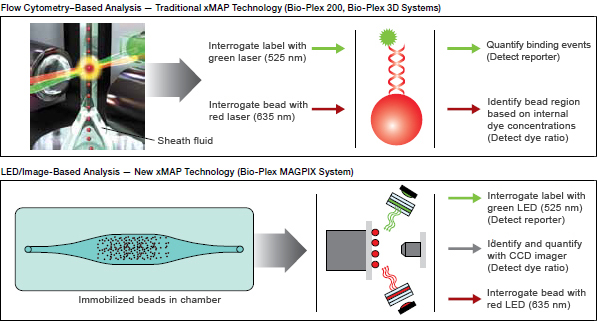
Fig 2. Data acquisition and reduction. Dyed beads are pushed through a detection chamber in a single file or magnetically immobilized. The red classification laser (635 nm) or LED interrogates the internal dyes to identify bead regions. The green reporter laser (532 nm) or LED interrogates the fluorescent reporter to measure analyte concentration.
Luminex xMAP technology is frequently compared to the traditional ELISA technique, which is limited by its ability to measure only a single analyte. The inability of traditional ELISA to multiplex presents additional limitations in sampling volume, cost, and labor (Figure 3). While the basic concepts of detection are similar between ELISA and Luminex xMAP technology, there are some important differences that center mainly on the capture antibody support. Unlike with traditional ELISA, Luminex xMAP capture antibodies are covalently attached to a bead surface, effectively allowing for a greater surface area as well as a matrix or free solution/liquid environment to react with the analytes. The suspended beads allow for assay flexibility in a singleplex or multiplex format.
Comparison of Bio-Plex Multiplexing vs. ELISA
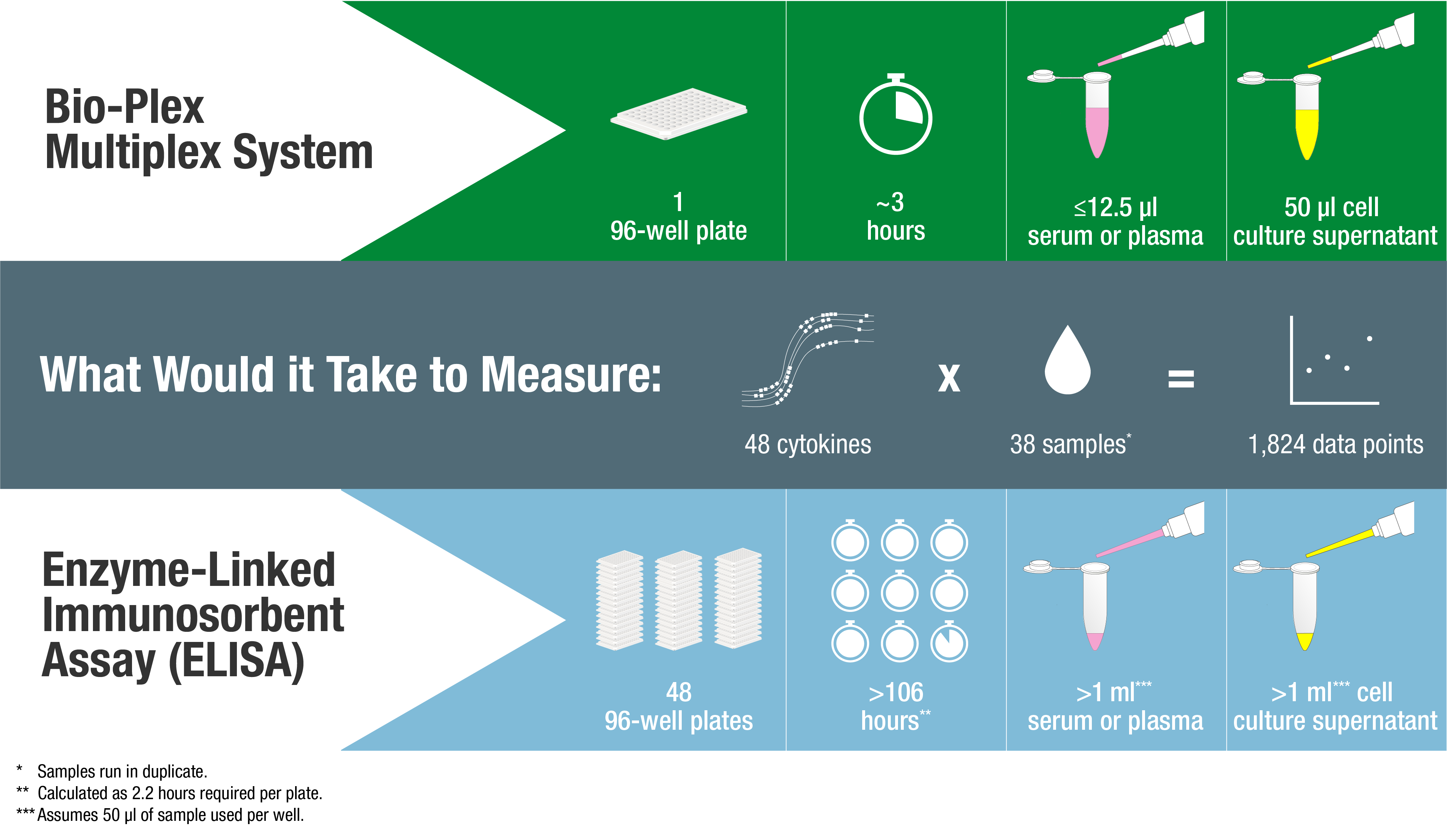
Fig 3. Protocol comparison between traditional ELISA and Bio-Plex system.
Bio-Plex Quick Tip Videos
-
Bio-Plex Quick Tips — Planning for Assay Success Part 1
-
Bio-Plex Quick Tips — Planning for Assay Success Part 2
-
Bio-Plex Quick Tips — Planning for Assay Success Part 3
Engvall E and Perlmann P (1971). Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 8, 871–874.
Leguin RM (2005). Enzyme immunoassay (EIA)/enzyme-linked immunosorbent assay (ELISA). Clin Chem 51(12), 2415–2418.
Van Weeman, BK and Schuurs AH (1971). Immunoassay using antigen-enzyme conjugates. FEBS Lett 15(3), 232-236.
Yalow RS and Berson SA (1960). Immunoassay of endogenous plasma insulin in man. J Clin Invest 39, 1157-1175.
Related Content
Videos
Dr Spinale, Professor of Cardiothoracic Surgery at Medical University of South Carolina, explains how the Bio-Plex system allowed him to tackle important questions in childhood disease research.
The Bio-Plex system allows researchers and clinicians to obtain precise results with less time and effort, using lower sample volumes in a high-throughput format.