- Target
Discovery - Lead
Discovery - Preclinical
Research - Clinical
Research - Process
Development
- Manufacturing
and Testing
The required production quantities of a candidate drug molecule increase greatly as the development cycle moves from discovery through development to approval and large-scale manufacturing. Design and optimization of production scale-up (including pilot plant operations), also known as process development, requires careful testing and monitoring using a wide range of different techniques including protein purification, analysis, quantitation, and identification tools at every step. Show more
One field of active research is to determine how small-scale models can be used to both design scale-up procedures and permit more rapid testing and optimization of process development. This is called scale-down, and provides greater flexibility for testing multiple parameters, more rapid system characterization, and iterative improvements. Furthermore, scale-down can be used for validation purposes. In addition to saving time, scale-down can significantly reduce costs. Sensitive analytical techniques are required for accurate evaluation of scale-down. Show less
Page Contents
|
|
Gene ExpressionDetect and quantitate nucleic acids in your scale-up/scale-down batches. Identify and measure any copurifying residual host cell DNA. Droplet Digital™ PCR Systems
|
|
|
Protein PurificationDevelop and finesse protein purification strategies across process development and scale-up using our extensive range of chromatography resins and instrumentation. NGC™ Medium-Pressure Chromatography Systems with IQ/OQ and CFR-Compliant Software |
|
|
Protein AnalysisUse our comprehensive protein analysis tools to monitor protein profiles throughout scale-up and validate antibodies used to detect and identify contaminating host cell proteins (HCP). Western Blotting Reagents and Equipment |
|
|
AntibodiesSave process development time with a wide selection of off-the-shelf antibodies. Utilize our custom antibody services for more specific antibody requirements, including for assay manufacturing purposes. Antibodies by Application |
Biochemical and Cellular Assays
In vitro and in vivo discovery approaches with high sample throughput and high-content analysis to characterize biological responses.
Biomarker Discovery & Validation
Biomarker monitoring across drug discovery, development, and clinical stages is key to defining drug effects.
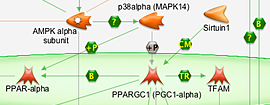
Biologic Development
Biologic development brings patients closer to personalized medicine to treat health problems.
Biosimilar Development
Biosimilar development requires physicochemical and functional characterization to ensure similarity.
Cancer Immunotherapy Development
Learn about trends in cancer immunotherapy development, such as CAR-T, and tools for genomic, cellular, and proteomic research.
CRISPR Gene Editing
CRISPR enables researchers to edit their desired target genes efficiently and accelerate drug development.
Viral Vector Vaccine Development
The COVID-19 pandemic opened the door to expedited vaccine development using new technologies and therapies that have traditionally been used in other applications.