Explore A1c and F/A2 testing with the D-10™ Hemoglobin Testing System
Experience the Compact, Easy-to-Use D-10 for Accurate Automated HbA1c and HbF/HbA2 Results
Ordering Information
For technical specifications and ordering information for the D-10 System and accessories, click here.
Explore
Frequently Asked Questions for D-10 Hemoglobin Testing Solutions
The Bio-Rad D-10 Hemoglobin Testing Solution is an ion-exchange HPLC to provide precise and reproducible results of hemoglobin binding proteins. It includes dual testing programs for HbA1c and fetal screening including HbA2, and HbF even in the presence of common hemoglobin variants such as HbS, HbC, HbD, and HbE. It is NGSP certified, IFCC traceable, and standardized to DCCT guidelines, ensuring compliance with international standards.
Yes. The D-10 HbA1c Assay is CE marked and FDA cleared as these certifications ensure the assay delivers accurate, reliable, and internationally standardized HbA1c results, including: NGSP certified (National Glycohemoglobin Standardization Program)1 IFCC traceable (International Federation of Clinical Chemistry and Laboratory Medicine)2 Standardized to DCCT guidelines, following American Diabetes Association (ADA) recommendations3
Yes. The D-10 is designed for ease of use with a compact footprint, minimal maintenance, and intuitive software. It installs quickly—often in a single service visit—and delivers walkaway operation, making it ideal for busy clinics and physician offices. Countries have clinical laboratory requirements that must be fulfilled to perform clinical diagnostic tests.
Yes, including: Built-in Software: The D-10 includes integrated software that streamlines system startup, automates quality control checks, and enables chromatogram export—reducing manual steps and improving consistency. Touchscreen Interface: An intuitive touchscreen makes navigation simple, enhancing ease of use for daily operation and data handling. LIS Integration: The D-10 supports connectivity with Laboratory Information Systems (LIS), allowing automatic data transfer for faster, more efficient workflows.
For the Bio-Rad D-10, routine operation includes a few daily checks, with minimal ongoing maintenance—only monthly and periodic replacement of the sample probe.
References:
Little RR et al. (2015). Effects of 49 Different Rare Hb Variants on HbA1c Measurement in Eight Methods. Journal of Diabetes Science and Technology I-8. https://pubmedncbi.nlm.nih.gov/25691657
DCCT Research Group (1996). The absence of a glycemic threshold for the development of long-term complications: the perspective of the Diabetes Control and Complications Trial. Diabetes 45(10), 1289–1298.
U.K. Prospective Diabetes Study (UKPDS) Group (1998). Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352:837-53. ADA (2022). Standards of medical care in diabetes – 2022. Diabetes Care, 45 (Supplement 1), S17-S38
Disclaimer:
The content on this website is for educational and informational purposes only and should not be considered medical advice or a recommendation for diagnosis or treatment. Always consult a qualified specialist when needed. Bio-Rad and contributors make no warranties regarding the accuracy or completeness of the information and disclaim all liability for its use. Users should review relevant updates and regulatory guidance.
These pages list our product offerings in these areas. Some products have limited regional availability. If you have a specific question about products available in your area, please contact your local sales office or representative.
D-10 Hemoglobin Testing Features

D-10 Rack Loader for Increased Efficiency
Keep pace with growing HbA1c testing demands by adding a D-10 Rack Loader. As diabetes rates rise, your lab’s testing volume may increase—but with the scalable D-10 System, there’s no need to invest in a new platform or repeat validation studies.
Provides convenient sample loading for walkaway capability
Brings enhanced flexibility with accommodate high-priority (STAT) sample processing
Offers 5x greater testing capacity than the standalone D-10 System
Boost walkaway capacity and streamline workflow with the D-10 Rack Loader—installed in a single service visit to enhance efficiency and productivity.
D-10 Dual Program: Simplify HbA1c and β-Thalassemia Testing with One Kit
Test for diabetes and β-thalassemia using a single kit with the D-10 Dual Program. Perform rapid, automated HbA1c analysis and in-depth β-thalassemia testing—then switch between methods instantly, with no need to change reagents or cartridges.
Short Method
Rapid HbA1c results in 3 minutes 400 HbA1c tests
Extended Method
HbA2, HbF, and HbA1c quantifiable results in 6.5 minutes HbA1c is reportable in the presence of heterozygous HbS, HbC, HbD, and HbE 400 HbA1c or 200 HbA2/F/A1c tests
Laboratory Experience with the D-10 Hemoglobin Testing System
Discover why clinicians choose the Bio-Rad D-10 Hemoglobin Testing System for their labs and clinics. Explore key features that matter most and how the system can enhance your diagnostic workflow.
Improving Anemia and Thalassemia Screening with the Bio-Rad D-10
Watch the video to hear what a key opinion leader says about the D-10 Dual Program HbA2/F/A1c.
Accordion text: I am Dr. Shanas Kaii, Consultant Hematopathologist at the PD Hinduja Hospital in Mumbai, India. I have been associated with Hinduja Hospital for over 30 years. My role is to oversee every function of the laboratory. As the first hospital-based lab in India accredited by the College of American Pathologists, we uphold the highest standards. I also serve as the President of the Indian Society of Hematology and Blood Transfusion.
Our lab specializes in hematology, making hemoglobinopathy testing and thalassemia screening essential. These tests are critical for diagnosing anemia and are part of our hemolytic anemia profile. Over the years, results from the Bio-Rad D-10 system have enabled accurate diagnoses, allowing for timely treatment.
Before adopting the D-10 system, we relied on alkaline and acid gel electrophoresis, which was tedious, limited, and prone to variant overlap common in India. When we looked to automate hemoglobinopathy screening, we considered two Bio-Rad systems. The VARIANT II was too advanced for our moderate workload, while the D-10 system was ideal. It effectively detects nearly all hemoglobin variants and thalassemias prevalent in India, making it integral to our lab.
We chose the D-10 for its accuracy and reliability. In a hospital-based lab, prompt treatment depends on fast, accurate diagnosis. The D-10 delivers results—from calibration to printout—in about six minutes, even faster than serum iron studies. This speed allows us to quickly confirm or rule out hemoglobinopathies as a cause of anemia.
We conduct around 85 tests per month for Hemoglobin A2 and F—about 1,000 annually. This number could be higher if we had obstetrics and pediatrics specialties. The D-10 also improves lab productivity with a 6-minute turnaround for thalassemia testing and 3 minutes for HbA1c. It runs small daily batches economically, eliminating delays from waiting to accumulate samples.
The staff finds the D-10 easy to use due to its full automation, eliminating the need for sample prep or manual data entry. It requires minimal maintenance, and the touchscreen interface with onboard computer simplifies kit changes. The D-10 handles our workload efficiently, even during staff shortages.
We appreciate features like sample and hemoglobin level alerts. Chromatogram review helps identify degraded samples—such as those with high bilirubin—allowing us to reprocess or dilute for accurate results. The hemoglobinopathy chromatogram also helps us incidentally identify elevated HbA1c levels. Quality control data is easily accessible and stored, with up to 10,000 results retained, reducing backup needs.
We value Bio-Rad’s quality products and consistent after-sales support. Their courteous staff is always ready to help. In our lab, the D-10 supports three key patient scenarios: health checkups with premarital screening, prenatal screening for pregnant women, and follow-up testing for patients with thalassemia-like CBC results.
The D-10 detects both common and rare beta and alpha chain variants found in India, providing a comprehensive hemoglobinopathy screening solution. We've identified variants such as double heterozygous for HbC and beta thalassemia, HbD Punjab homozygous, sickle cell with hereditary persistence of fetal hemoglobin, and delta-beta thalassemia. We've also detected cases like homozygous thalassemia with high HbF, hemoglobin H, hemoglobin J (eluting in P3), and hemoglobin Q India (at 4.4 minutes).
Bio-Rad’s online Library of Hemoglobin Variants helps us interpret abnormal chromatograms and report with confidence. Bio-Rad also supports clinical education through partnerships like with the Thalassaemia International Federation, offering programs like TIF certification and “Test Your Knowledge.”
We’ve also used the D-10 in research, including a study on borderline Hemoglobin A2 values (3.5–3.9%) with DNA mutation analysis to detect silent mutations. We identified variants such as IVS-I-5 G>C and IVS-I-1 G>T.
In summary, the D-10 is compact, easy to use, and employs gold-standard ion-exchange HPLC for trusted, accurate results. It’s a workhorse that handles increased workloads without compromising quality—an ideal choice for small to medium-sized labs offering both hemoglobin A2/F and HbA1c testing on one platform with a single kit.
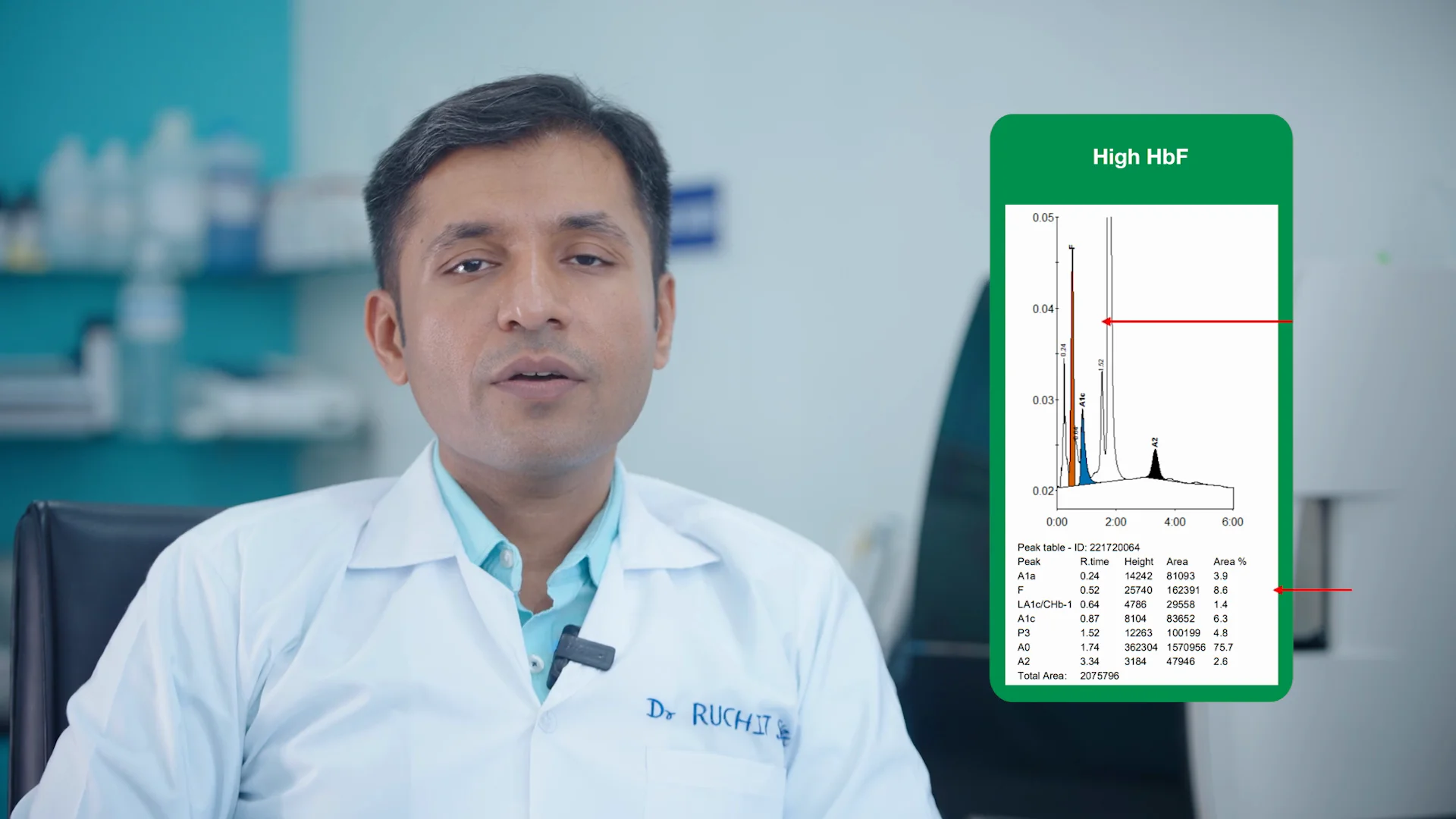
Inside the Lab: How the D-10 Streamlines Walk-In Patient Testing
Watch the video to see how a laboratory director uses the D-10 System’s dual programs to streamline testing in a walk-in patient lab.
Dr. Ruchit Shah M.D., Laboratory Director, Consultant Pathologist Mangalam Pathology Laboratory in Ahmedabad, India
Increased Productivity
Discover in this video how the D-10 System streamlines lab workflow—eliminating sample prep, reducing maintenance, and removing the need for manual data entry. With an intuitive touchscreen and onboard computer, it’s built for ease of use, saving time and increasing productivity.
“The D-10 rapidly changed the way we test.”
Jamie Justice, Quality Manager Bossier Family Medicine
Cost Efficiency
See how the D-10 System drives efficiency and value in your lab—delivering high-quality, automated results with no hidden costs. Watch the video to learn how one laboratory streamlined operations and cut expenses using Bio-Rad’s trusted ion-exchange HPLC technology.
“The D-10 is 'rock solid' and makes economic sense.”
Matt Wheeler, Administrator Bossier Family Medicine
Testing for HbA1c in an Endocrine Practice
In this video, see how a diabetes clinic uses dual D-10 instruments to deliver real-time A1C results with minimal staff effort. And understand how the D-10 improves testing outcomes.
“We have chosen the D-10 System because of its accuracy and reliability.”
Dr. David Robertson M.D., Endocrinology Atlanta Diabetes Associates
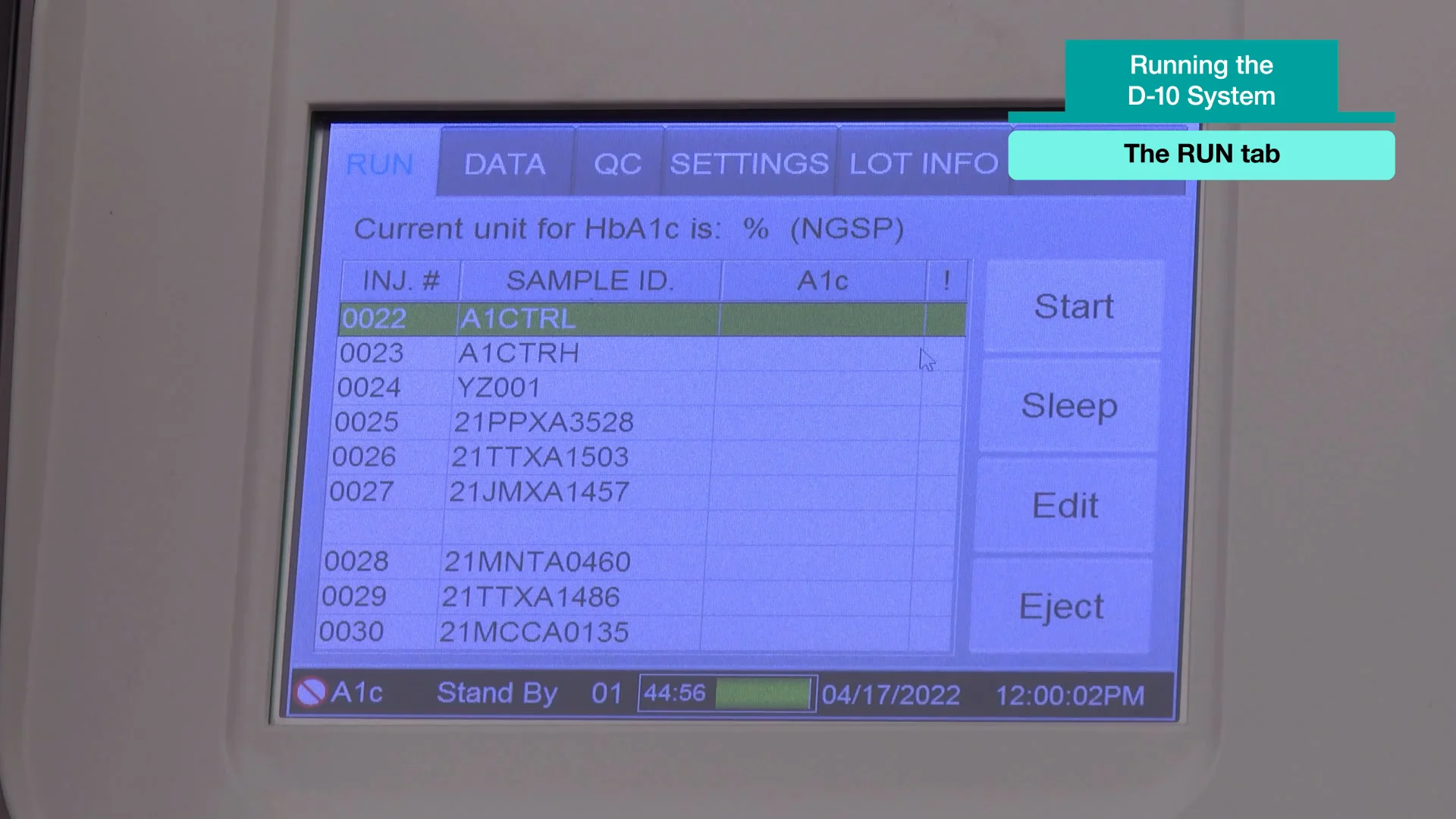
See How Simple It Is to Operate the D-10 System – Watch the Videos
Discover how simple and efficient it is to operate the Bio-Rad D-10 Hemoglobin Testing System—perfect for clinical labs and physician offices. Just power on the compact analyzer, perform a quick system check, and load the ready-to-use reagent cartridge and buffer pack. Scan patient or QC information, insert the sample, and choose your testing method. The D-10 delivers fast, NGSP-certified HbA1c results with built-in QC tracking, automated result storage, and low-maintenance operation—streamlining your workflow and ensuring reliable diabetes monitoring.
How Do Other Systems Compare to the D-10 System When Rare, Abnormal Hemoglobin Variants are Present?
Effects of 49 Different Rare Hb Variants on HbA1c Measurement in Eight Methods
Randie R. Little et al. (2015) Journal of Diabetes Science and Technology¹
Bio-Rad HPLC systems outperformed other HbA1c testing systems1. With the D-10 System, 100% clinically accurate results in the presence of rare hemoglobin variants would have been reported. The table below illustrates the clinical benefit of Bio-Rad ion-exchange HPLC for patient diagnosis and treatment.
| Total samples | % Total samples with accurate results | Instrument | Method | # Variants Tested | # Variants that interfered with A1c results | % Variants that interfered with A1c results |
|---|---|---|---|---|---|---|
| 75 | 100% | Bio-Rad D-10 System | HPLC | 39 | 0 | 0% |
| 84 | 97.7% | Bio-Rad VARIANT II TURBO System | HPLC | 43 | 2 | 4.6% |
| 87 | 96.6% | Tosoh G7 | HPLC | 43 | 2 | 6.9% |
| 88 | 92.1% | Roche Tinaquant | Immunoassay | 44 | 5 | I11.3% |
| 33 | 87.9% | Sebia Capillarys 2 | Capillary electrophoresis | 18 | 2 | 11.1% |
| 88 | 87.5% | Diazyme Direct Enzymatic | Enzymatic | 44 | 8 | 18.1% |
| 87 | 67.7% | Tosoh G8 | HPLC | 44 | 9 | 20.4% |
Read Case Study about variant II turbo 49 variant
References:
1. Little RR et al. (2015). Effects of 49 different rare Hb variants on HbA1c measurement in eight methods. J Diabetes Sci Technol 9(4), 849-856. https://pubmed.ncbi.nlm.nih.gov/25691657.
What are the Benefits to D-10 Hemoglobin Testing Solutions?

D-10: Compact Design
Entire platform sits within a 41” width x 54” length x 50” height with an integrated computer to save valuable bench space.
Low Maintenance, Fewer Service Visits
Maximize productivity with minimal maintenance requirements—just routine buffer and column checks—reducing technician workload and minimizing unexpected downtime with fewer than one service visit per year.
Easy Use and Training
The D-10 System is widely adopted for its ease of use and quick staff training—automated features, simple reagent prep, and a multilingual interface streamline HPLC testing and ensure smooth simplified results review integration into any lab workflow.