Hydrophobic interaction chromatography (HIC) separates molecules based on their hydrophobicity. HIC is a useful separation technique for purifying proteins while maintaining biological activity due to the use of conditions and matrices that operate under less denaturing conditions.
This section provides an overview of hydrophobic interaction chromatography with general considerations about various factors such as the ligand, matrix, salt concentration, pH, and temperature.
Related Topics: Affinity Chromatography, Size Exclusion Chromatography, Ion Exchange Chromatography, Mixed-Mode Chromatography, Low Pressure Chromatography Systems and Medium Pressure Chromatography Systems.
Page Contents
The principle for protein adsorption to HIC media is complementary to ion exchange and size exclusion chromatography. Sample molecules containing hydrophobic and hydrophilic regions are applied to an HIC column in a high-salt buffer. The salt in the buffer reduces the solvation of sample solutes. As solvation decreases, hydrophobic regions that become exposed are adsorbed by the media. The more hydrophobic the molecule, the less salt is needed to promote binding. Usually a decreasing salt gradient is used to elute samples from the column in order of increasing hydrophobicity. Sample elution may also be assisted by the addition of mild organic modifiers or detergents to the elution buffer.
Bio-Rad provides media for hydrophobic interaction chromatography such as Macro-Prep® HIC media and Bio-Beads™ SM-2 media.
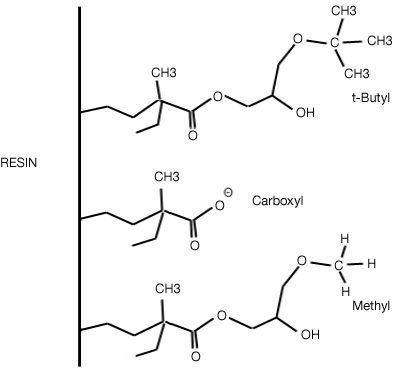
Macro-Prep HIC media. Note: Macro-Prep t-butyl contains t-butyl and carboxyl groups.
This section provides general considerations for hydrophobic interaction chromatography including factors such as the ligand, matrix, salt concentration, pH, and temperature.
Ligand
A protein's adsorption behavior is determined by the type of immobilized ligand. In general, straight chain alkyl ligands demonstrate hydrophobic character while aryl ligands show a mixed mode behavior where both aromatic and hydrophobic interactions are possible (Hofstee and Otillio, 1978). The choice of ligand type is empirically determined.
Degree of substitution
The protein binding capacity increases with an increased degree of substitution of the immobilized ligand. With a high level of ligand substitution, the binding capacity remains constant; however, the affinity of the interaction increases (Jennissen and Heilmeyer, 1975). Proteins bound under these conditions are difficult to elute due to multi-point attachment (Jennissen, 1978).
Matrix
The most widely used supports are hydrophilic carbohydrates: cross-linked agarose and synthetic copolymer materials. The selectivity between different supports will not be identical though the ligands may be the same. Modify adsorption and elution conditions to achieve similar results when moving from one media to another.
Salt concentration
The addition of structured salts to the equilibration buffer and sample promotes ligand-protein interactions in HIC (Porath et al.,1973). As the salt concentration increases, the amount of bound protein increases as does the risk of protein precipitation at the higher ionic strength.
The figure below represents the Hofmeister series on the effect of some anions and cations on protein precipitation. Though sodium, potassium or ammonium sulphates produce relatively higher precipitation effects, these salts effectively promote ligand-protein interactions in HIC. Most bound proteins are eluted by washing with water or dilute buffer at near neutral pH.

Effect of anions and cations on protein precipitation.
pH
HIC mobile phases are typically in the neutral pH range from 5–7 and buffered with sodium or potassium phosphate. In general, the strength of the interaction between proteins and the media decreases with increasing pH as a result of increased charge of the protein due to the titration of acidic groups. This effect can vary from protein to protein. Thus, pH can impact the level of protein binding and the selectivity of the media. However, changes in pH do not have a significant effect over moderate ranges. Though it is useful to determine the optimal pH, pH gradients are not generally used as an elution method.
Temperature
The affinity of hydrophobic interactions increases with temperature. Temperature also impacts protein structure, solubility, and the interaction with the HIC matrix. Because temperature effects can be difficult to predict, it is generally not used to modulate separation using HIC. Not surprisingly, experiments conducted at room temperature may not be reproduced in a cold room.
| Suitability** | |||||
| Media Type | Packaging Format* |
Analytical Scale |
Pilot/ Preparative Scale |
Process Scale |
Application |
| Hydrophobic Interaction | |||||
| Macro-Prep® Methyl | B | ++++ | ++++ | + | Separation of proteins based on relative hydrophobicity; pH stability 1–10 |
| Macro-Prep® t-butyl | B | ++++ | ++++ | + | Separation of proteins based on relative hydrophobicity; pH stability 1–10 |
* B, bottle; C, cartridge (1 ml or 5 ml); GC, gravity column; SC, spin column; HPLC, high-pressure column; MPC, medium-pressure column.
** +, low suitability; ++, moderate suitability; +++, suitable; ++++, high suitability.
Hofstee BH and Otillio NF (1978). Non-ionic adsorption chromatography of proteins. J Chromatogr 159, 57–69. PMID: 649758
Jennissen HP (1978). Multivalent interaction chromatography as exemplified by the adsorption and desorption of skeletal muscle enzymes on hydrophobic alkyl-agaroses. J Chromatogr 159, 71–83. PMID: 418077
Jennissen HP and Heilmeyer LM, Jr. (1975). General aspects of hydrophobic chromatography. Adsorption and elution characteristics of some skeletal muscle enzymes. Biochemistry 14, 754–760. PMID: 163642
Porath J et al. (1973). Salting-out in amphiphilic gels as a new approach to hydrophobic adsorption. Nature 245, 465–466. PMID: 4356152
