
Visit This Page Often for Valuable Teaching Resources
This section of our website provides educators with resources created for our hands-on, inquiry-driven Biotechnology Explorer workshop series. The PowerPoint presentations and laboratory quick guides are available for educational use only. We will continue to add additional teaching tools and resources to this page. Use these materials in your classrooms and teaching labs as you see fit.
|
Animated tutorials for ELISA, PCR, and interactive DNA crime lab Grant money (for US teachers only) pGLO DNA sequence download Three-dimensional (3-D) structure of Green Fluorescent Protein (GFP).
|
For additional information and imagery that involve the unique properties of GFP, we recommend that you visit the website developed and maintained by the Center for BioMolecular Modeling at the Milwaukee School of Engineering: GFP 3-D images.
PowerPoint Presentations for Workshops and for Teaching
Back to top
Grant Money — For Teachers in the U.S. Only
Where and How to Obtain Grant Money for Biotechnology in Your Classroom or District

"In a world where everything is becoming 'virtual,' grants are the real thing. They have helped our staff overcome a shortage of resources and transform a vision into a reality. On our campus, over 500 students a year experience the world of biotechnology through programs supported by grants."
— Stan Hitomi, Coordinator, Math and Science, San Ramon Valley Unified School District, Danville, CA
Many thanks to Stan Hitomi for helping assemble this exceptional guide to understanding the grant-writing process.
Did You Know?
Concepts important to biotechnology are dealt with in various degrees of specificity and comprehensiveness in the new National Science Education Standards. If your curriculum decisions are based on these standards, biotechnology will receive unprecedented emphasis in your science classroom.
The Burning Question: How Do You Secure the Funds to Pay for It?
Teachers are creative and energetic professionals, with their own ideas about how to implement the new National Science Education Standards and how to integrate classroom and district-wide projects that will enhance their students' learning experience. Many schools have limited funding and are unable to support such projects. In the absence of district or site funding, many teachers have turned to grants to make their projects become a reality.
Grants Are a Highly Accessible Source of Funding
Educational grants come in all sizes, from small local grants (hundreds to tens of thousands of dollars) to state and national grants (tens of thousands to millions of dollars). The funding sources for grants vary from private businesses and associations to state and national organizations and foundations. Applications for these funds range from a simple letter to a formal comprehensive application package. The key is to select the scope and source of funding that most closely meets the needs of your program.
The number of grants available to teachers has grown dramatically in recent years, especially in the areas of science and technology. In addition, a great deal of grant money is left unclaimed every year due to a lack of applicants.
Go for It: Write a Grant!
In preparation for writing a grant or seeking the assistance of a grant writer, it is best to consider the following major components of a typical grant application.
Summary: One of the most important pieces of the grant application, this brief abstract of the project provides readers with a first impression. It is best to prepare this section after the proposal has been developed. This will ensure that all the key components of the proposal are included and coherent. The summary can also be used to keep administrators, department members, and the media informed about your project.
Introduction: This section describes your school, district, or organization and the community that you serve. Make certain to identify the people who will be served by the project and the significance of the project to them. Include a biographical sketch, resume, or curriculum vitae of the primary participants in the project. Also include the goals and philosophy of the organization represented in the proposal.
Problem Definition and Need: This statement must be clear, concise, and supported by data whenever possible. The statement should clearly describe a need as it relates to your students and/or teachers. It should be clear from this section how the problem will be overcome through the funding. For each problem presented, there must be a corresponding goal and objective.
Goals and Objectives: Your goals and objectives should be clearly defined. It is important to link goals and objectives to the specific criteria of the funding program.
Methods and Activities: This section outlines the tasks, who will do them, when they will be completed, and what resources will be needed. It is important to be as specific as possible; for example, identify people or positions assigned to each task, state the number of students to be served, etc. A timeline graphic is highly recommended.
Evaluation: Describe how the success of the program will be measured. List the data to be collected and the process used to evaluate them. Delineate a plan for measuring progress throughout the funding period and say how modifications will be made.
Budget: Outline expenses in detail here. It is often possible to obtain a free quote for equipment and supplies from vendors to assist in completing this section (Bio-Rad is happy to provide this assistance). It is helpful, and often required, to break down budgets into categories such as personnel, travel, equipment, and supplies. The budget must be consistent with the methods and activities of the project.
Sustainability: Funding is often based upon the ability of a project to succeed beyond the grant. It is helpful to indicate how the program will be sustained beyond the funding cycle. Include comments about the maintenance, repair, and upgrade of equipment. Also include any plans to disseminate or expand the program beyond the scope described in the proposal.
Grant-Writing Information
| Grantwriting (Corporation for Public Broadcasting) |
Back to top
Grantors and Directories
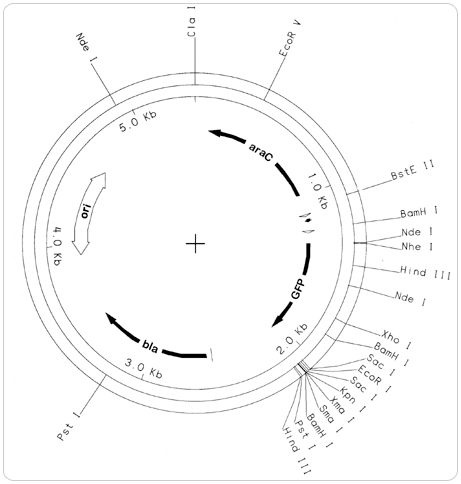
pGLO Plasmid Map
Our unique pGLO plasmid map is available for educational use only.
| Download pGLO DNA Sequence (Microsoft Word document) |
pGLO contains several genes that enable replication of the plasmid DNA and expression of the fluorescent trait (phenotype) in bacteria following transformation. Some of the essential genes include:
GFP — The jellyfish gene that codes for the production of Green Fluorescent Protein.
bla — A gene that encodes the enzyme beta-lactamase, which breaks down the antibiotic ampicillin. Bacteria containing the bla gene can be selected by placing ampicillin in the growth medium.
ori — The origin of pGLO plasmid DNA replication.
araC — A gene that encodes the regulatory protein that binds to the pBAD promoter. Only when arabinose binds to the araC protein is the production of GFP switched on.
pBAD Promoter — A specific DNA sequence upstream from the GFP gene, which binds araC-arabinose and promotes RNA polymerase binding and transcription of the GFP gene.
Multiple Cloning Sites — Regions of known restriction sites (NdeI, HindIII, EcoRI, etc.) that permit insertion or deletion of the gene of interest.
To learn about the process used to generate this unique fluorescence, please visit www.maxygen.com.
pGLO Gene Regulation. One Gene, One Protein?
Our bodies contain thousands of different proteins, which perform many different jobs. Digestive enzymes, some of the hormone signals that run through our bodies, and the antibodies protecting us from disease are all proteins. The information for assembling a protein is carried in our DNA. The section of DNA that contains the code for making a protein is called a gene. There may be up to 100,000 genes in the human genome. Each gene codes for a unique protein: one gene, one protein. The gene that codes for a digestive enzyme in your mouth is different from one that codes for an antibody or a pigment that colors your eyes.
Organisms regulate the expression of their genes, and ultimately, the amounts and kinds of proteins present within their cells, for a myriad of reasons including development, cellular specialization, and adaptation to the environment. Gene regulation not only allows adaptation to differing conditions but also prevents wasteful overproduction of unneeded proteins, which would put the organism at a competitive disadvantage. The genes involved in the transport and breakdown (catabolism) of food are good examples of highly regulated genes. For example, the sugar arabinose is both a source of energy and a source of carbon. E. coli bacteria produce three enzymes (proteins) needed to digest arabinose as a food source. The genes that code for these enzymes are not expressed when arabinose is absent, but they are expressed when arabinose is present in their environment. How is this so?
Regulation of protein expression often occurs at the level of transcription from DNA into RNA. This regulation takes place at a very specific location on the DNA template, called a promoter, where RNA polymerase binds the DNA and begins transcription of the gene. In bacteria, groups of related genes are often clustered together and transcribed into RNA from one promoter. These clusters of genes controlled by a single promoter are called operons.
The three genes (araB, araA, and araD) that code for the three digestive enzymes involved in breaking down arabinose are clustered together in what is known as arabinose operon 3. These three proteins depend on initiation of transcription from a single promoter, PBAD. Transcription of these three genes requires the simultaneous presence of the DNA template (promoter, gene, and operon), RNA polymerase, a DNA binding protein called araC, and arabinose. The araC protein binds to the DNA at the binding site for RNA polymerase (the beginning of the arabinose operon).
When arabinose is present in the environment, bacteria take it up. Once inside, the arabinose interacts directly with araC, which is bound to the DNA. The interaction causes araC to change shape, which in turn helps RNA polymerase bind the promoter, and the three genes araB, araA, and araD are transcribed. Three enzymes are produced; they do their job to break down arabinose, and eventually the arabinose runs out.
The DNA code of the pGLO plasmid has been engineered to incorporate aspects of the arabinose operon. Both the promoter (PBAD) and the araC gene are present. However, the genes that code for arabinose catabolism, araB, araA, and araD, have been replaced by the single gene that codes for the Green Fluorescent Protein (GFP). Therefore, in the presence of arabinose, araC protein promotes the binding of RNA polymerase, and GFP is produced. Cells fluoresce a brilliant green as they produce more and more protein. In the absence of arabinose, araC no longer facilitates the binding of RNA polymerase, and the GFP gene is not transcribed. When the GFP protein is not made, bacteria colonies will appear to have a wild-type (natural) phenotype — of white colonies with no fluorescence. This is an excellent example of the central molecular framework of biology in action: DNA > RNA > PROTEIN > TRAIT.
Back to top

Giant 40" Biotech Universe Wall Poster — Bulletin Number 2370
This beautiful and colorful poster is also a fabulous teaching aid designed to highlight the central molecular framework of biology and generate discussion about the flow of biological information from DNA > RNA > PROTEIN > TRAIT. It's free — just call 1-800-876-3425, ext. 1, or if you are a registered user, order online via your literature folder.
Back to top

