Polyacrylamide is ideal for protein separations because it is chemically inert, electrically neutral, hydrophilic, and transparent for optical detection at wavelengths greater than 250 nm. Additionally, the matrix does not interact with the solutes and has a low affinity for common protein stains. This section provides an overview of the properties and characterization of polyacrylamide gels, the advantages and disadvantages of precast vs. handcast gels, and examples of migration charts.
Related Topics: Protein Standards, Buffer Systems and Gel Chemistries, and Handcasting Polyacrylamide Gels.
Page Contents
Polyacrylamide gels are prepared by free radical polymerization of acrylamide and a comonomer crosslinker such as bis-acrylamide. Polymerization is initiated by ammonium persulfate (APS) with tetramethylethylenediamine (TEMED) as the catalyst (see figure below). Riboflavin (or riboflavin–5'–phosphate) may also be used as a source of free radicals, often in combination with TEMED and APS. Polymerization speed depends on various factors (monomer and catalyst concentration, temperature and purity of reagents) and must be carefully controlled since this process generates heat and polymerization that if too rapid may lead to non-uniform pore structures.
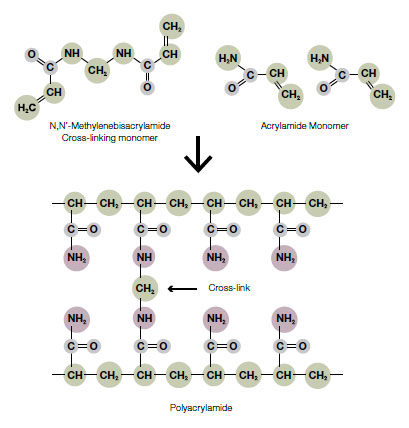
Polymerization of acrylamide monomers and bis-acrylamide.
Polyacrylamide gels are characterized by two parameters: total monomer concentration (%T, in g/100 ml) and weight percentage of crosslinker (%C). By varying these two parameters, the pore size of the gel can be optimized to yield the best separation and resolution for the proteins of interest. %T indicates the relative pore size of the resulting polyacrylamide gel: higher %T refers to a larger polymer-to-water ratio and on average smaller pores.

The practical ranges for monomer concentration are stock solutions of 30-40%, with different ratios of acrylamide monomer to crosslinker. The designations 19:1, 29:1 or 37.5:1 on acrylamide/bis solutions represent crosslink ratios 5%, 3.3% and 2.7% (the most common crosslinker concentration for protein separations).
Gels can be made with a single, continuous percentage throughout the gel (single-percentage gels), or they can be cast with a gradient of %T through the gel (gradient gels). Typical gel compositions are between 7.5 and 20% for single-percentage gels, and typical gradients are 4–15% and 10–20%. Use protein migration charts and tables to select the gel type that offers optimum resolution of your sample (see figure below).
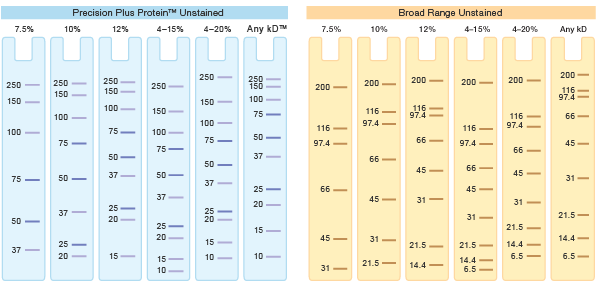
Examples of migration charts.
- Use single-percentage gels to separate bands that are close in molecular weight. Optimum separation occurs in the lower half of the gel, so choose a percentage in which your protein of interest migrates to the lower half of the gel
- Use gradient gels to separate samples containing a broad range of molecular weights. Gradient gels allow resolution of both high- and low-molecular weight bands on the same gel. The larger pore size towards the top of the gel permits resolution of larger molecules, while pore sizes that decrease toward the bottom of the gel restrict excessive separation of small molecules
- For new or unknown samples, use a broad gradient, such as 4–20% or 8–16%, for global evaluation of the sample, and then move to using an appropriate single-percentage gel once a particular size range of proteins has been identified
Precast gels are ready to use and offer greater convenience, more stringent quality control, and higher reproducibility than handcast gels. Many precast gels also provide a shelf life of up to 12 months, allowing gels to be stored and used as needed (this is not possible with handcast gels, as they degrade within a few days).
Handcast gels, on the other hand, must be prepared from acrylamide and bis-acrylamide monomer solution; the component solutions are prepared, mixed together, and then poured between two glass plates to polymerize (see Handcasting Polyacrylamide Gels for detailed protocols). Acrylamide and bis-acrylamide are neurotoxins when in solution, so care must be taken to avoid direct contact with the solutions and to clean up any spills. In addition, the casting process requires hours to complete and is not as controlled as it is by gel manufacturers, so handcast gels are more irregular and less reproducible.
Though handcasting offers the benefit of customized percentages, chemistries, and gradients, precast gels are sized to fit specific electrophoresis cells and are available in a range of chemistries, formulations, comb types, and thicknesses. Precast gels differ from their handcast counterparts in that they are cast with a single buffer throughout. Bio-Rad's precast gels also do not contain SDS. For a complete and current list of available precast gels, visit our Mini-PROTEAN precast gels or Criterion precast gels pages.
The size format of the gel used depends on the electrophoresis cell selected (see Protein Electrophoresis Equipment). Precast gels are available for Bio-Rad's mini- and midi-format electrophoresis systems, and handcasting accessories are available to fit all Bio-Rad electrophoresis cells.
Another parameter to consider is the number of wells and thickness of the gel, which depend on the number and volume of samples to analyze. To create sample wells in a gel, a comb is placed into the top of the gel prior to polymerization. When the comb is removed, a series of sample wells is left behind. The number and size of these wells dictate how many samples and of what volume may be loaded (see table below). The thickness of the gel also plays a role in determining the sample volume that can be loaded.
A variety of comb types are available for handcasting — refer to Mini-PROTEAN Tetra handcast systems for more information. The following comb types are available for precast gels.
| Comb Thickness, 1.0 mm | ||
| Number of Wells | Well Volume | |
| Mini-Format Gels (Ready Gel® and Mini-PROTEAN) |
8+1 | 30 µl |
| 10 | 30 µl and 50 µl | |
| 12 | 20 µl | |
| 15 | 15 µl | |
| IPG | 7 cm IPG strip | |
| Midi-Format Gels (Criterion) |
12+2 | 45 µ |
| 18 | 30 µl | |
| 26 | 15 µl | |
| Prep+2 | 800 µl | |
| IPG+1 | 11 cm IPG strip | |
