
Explore how to boost electro-transformation efficiency in mycobacteria. In this foundational study, Cirillo et al. use the Bio-Rad Gene Pulser™ System to demonstrate electro-transformation of Mycobacterium smegmatis. You’ll see how tuning factors such as culture density, pulse length, time constant, and preharvest incubation can significantly influence transformation efficiency, offering practical insights for working with slow-growing or hard-to-transform bacterial species. Read on to see how the workflow was optimized.
Featured Content
Jeffrey D. Cirillo*, Torin R Weisbrod, and William R. Jacobs, Jr. Howard Hughes Medical Institute, Department of Microbiology and Immunology, Albert Einstein College of Medicine, 1300 Morris Park Avenue, Bronx, NY 10461. *Current address: Stanford University School of Medicine, Department of Microbiology and Immunology, Shennan Fairchild Science Building# D309, Stanford, CA 94305
Introduction
Electroporation has been shown to be a useful technique for the transformation of a wide variety of prokaryotic and eukaryotic organisms. It was for this reason that attempts were made to apply this technique to mycobacteria. By doing so, we hoped to increase the transformation efficiency beyond that which could be obtained by spheroplast transformation, the technique which had been used previously. Initially, our attempts at obtaining transformants in Mycobacterium smegmatis using plasmids were not very successful, yielding approximately 10 transformants/µg of plasmid DNA2. The isolation of an M. smegmatis mutant, mc²155, in which plasmids were more stably maintained allowed us to obtain an increased efficiency of 104 transformants/µg with electroporation. Using information obtained from an in-depth study of factors affecting electro-transformation efficiencies of E. coli, we were able to re-evaluate our electroporation methods for M. smegmatis. It was found that use of low-salt electroporation buffers and 0.2 cm electrode gap cuvettes enabled us to apply higher field strengths and longer time constants to the sample. These refinements allowed the development of more efficient techniques for electroporation of M. smegmatis as well as the slow-growing mycobacteria, bacille Calmette-Guérin. Here we describe additional experiments designed to determine the electrical and biological parameters which have an effect upon the transformation efficiency of mycobacterial cells. These parameters include culture density, time constant, and pre-harvest incubation time on ice. Variations in these parameters are shown to have significant effects on the transformation efficiency of mycobacteria. Analysis of these data led to the proposal of conditions that may further improve the efficiency of transformation of M. smegmatis.
Materials and Methods
All experiments described were conducted using the high-efficiency transformation strain M. smegmatis mc2155. Cells were grown in M-ADC-TW broth. Preparation of electroporation-competent M. smegmatis cells was accomplished as described previously with the modifications indicated here. The cells were harvested at an A600 of 0.8 to 1.0, incubated on ice for approximately 2 hours, then washed with 10% glycerol three times (refer to Table 1 for the volume of the washes), and resuspended in 10% glycerol at 1/500th the original volume. The plasmid pMV262 (4489 bp), purified on a cesium chloride gradient, was used for all transformations. This plasmid carries an aminoglycoside phosphotransferase gene (aph) which allows selection for transformants on M-ADC plates containing 10 µg/ml kanamycin. Concentrations of pMV262 were determined by A260 readings and confirmed by ethidium bromide stained agarose gels. Ten ng of supercoiled pMV262 DNA were mixed with 100 µl of mycobacterial cells and placed in a 0.2 cm Bio-Rad cuvette. The Gene Pulser® apparatus and Pulse Controller accessory (Bio-Rad Laboratories) were used for all electroporation experiments at 25 µF and 2.50 kV. All electro-transformations were done in duplicate. Transformants were plated in duplicate on M-ADC plates and average transformation efficiencies were determined.
Results
The three parameters that we found important in influencing electrotransformation efficiencies of M. smegmatis are cell culture density, pulse length, and pre-harvest incubation at 0 °C.
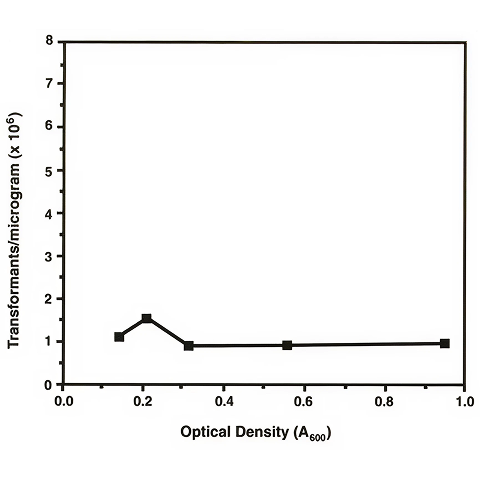
To determine the effects of culture density on transformation efficiency, a single one-liter culture was grown to an A600 of approximately 0.15. Aliquots of 100 ml were taken at various times and were incubated for 2 hours on ice before preparing electroporation-competent cells. In order to prevent the concentration of cells from having an effect upon the transformation efficiency, the cells were suspended in different volumes for each time point but at the same concentration of bacterial cells. Thus, the data obtained should be a measure of the effects of the stage of growth of the culture rather than of the number of cells present. A 15-25 msec time constant (25 µF and 1,000 Ω parallel resistor of the Pulse Controller) was used for each of these electroporation experiments. Figure 1 shows the average number of transformants obtained per µg of DNA at each time point.
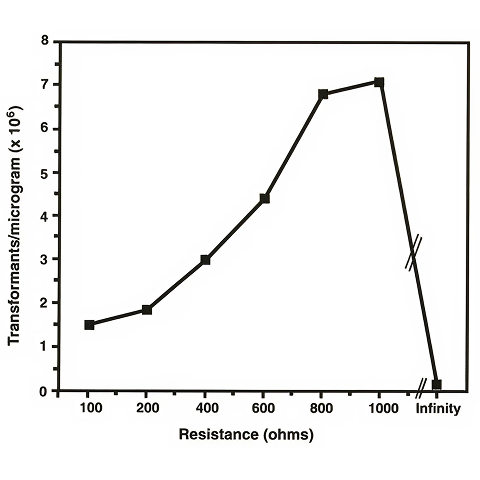
The effect of varying the pulse length was also studied. The Pulse Controller accessory to the Gene Pulser apparatus allows the use of an adjustable parallel resistor. This parallel resistor offers control of the time constant by allowing most of the current to pass through it rather than through the electrically resistant sample. The choice of the resistor, and therefore the time constant, has been shown to be important in affecting the efficiency of electroporation. Figure 2 shows the results of electroporation using different parallel resistors of the Pulse Controller ranging from 100 Ω to 1,000 Ω. The "infinity" setting indicates that no resistor is placed in parallel with the sample so that all of the current passes through the sample. The cells which were used for this experiment were harvested at an A600 of 0.8-1.0.
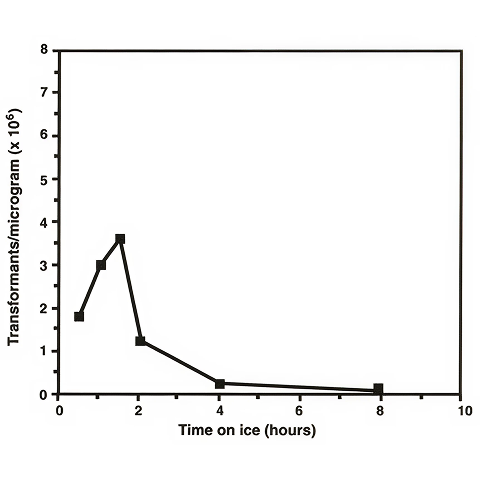
We found that the time the cells are allowed to incubate on ice between removal from the incubator and preparation for electroporation had an effect on electro-transformation efficiencies of mycobacteria. Dower et al. reported that E. coli cells were placed on ice for only 5 minutes before they were harvested by centrifugation. In the case of M. smegmatis, however, we observed that higher transformation efficiencies were obtained when the cells were allowed to incubate on ice for longer than 1 hour. In order to determine the optimum incubation time on ice, we harvested 100 ml time points from a 1 liter culture which had been placed on ice at an A600 = 0.8. The aliquots were individually prepared for electroporation. The results are shown in Figure 3.
Discussion
Careful attention to the various factors that influence the electro-transformation of M. smegmatis may greatly impact the transformation efficiency. Based on work reported on E. coli, we expected the density of the M. smegmatis culture to have a significant effect upon the transformation efficiency. In fact, we found the effect to be very small, at least in the logarithmic growth phase we studied. The concentration of viable E. coli cells at the time of electroporation has been reported to be one of the most important factors influencing the transformation efficiency. From the data obtained with mycobacteria, it can be concluded that there is no difference in the efficiency with which these cells can be transformed through early to late log phase growth. If electro-transformation is dependent on the number of viable cells present in the cuvette and the cells are suspended at the same concentration at each time point during the growth of the culture, the proportion of viable to non-viable cells must be approximately the same at each time point. Although these experiments did not test stages of growth other than log phase, it is likely that stationary phase and lag phase cells would not transform at nearly the same frequencies as cells present in log phase due to the lower percentage of viable cells present.
The time constant, varied by choosing different parallel resistors of the Pulse Controller, did appear to have a significant effect on the transformation efficiency of mycobacterial cells. The time constant that gave the highest transformation efficiencies, nearly 105 transformants/µg, was 15-25 msec (1,000 Ω and 25 µF). Use of the 1,000 Ω resistor allows a higher current to flow through the sample. Such high levels of current passing through an E. coli cell sample would result in killing such a large percentage of the cells that the apparent transformation efficiency would be very low. The fact that M. smegmatis can survive such high levels of current illustrates the hardy nature of mycobacterial cells, a characteristic that has thwarted earlier attempts at determining an efficient transformation technique for this genus. At the "infinity" setting on the Pulse Controller, the transformation efficiency is very low. The time constants at this setting are extremely long (in the range of several seconds), indicating that the current flows solely through the extremely resistant sample (no resistor in parallel). Although it appears that mycobacteria can withstand high voltages for long periods of time, the mycobacterial cells are probably killed by exposure to a pulse of such a long duration. A significant reduction of the number of viable cells would result in the lower transformation efficiencies observed.
A third aspect of electro-transformation which has a significant effect upon the transformation efficiency of mycobacteria is the length of time that the cells are incubated on ice before preparation. It is not understood why the amount of time that the bacterial cells are incubated on ice affects the transformation efficiency. However, this phenomenon has also been observed in E. coli where incubation of approximately 5 minutes on ice contributes to efficiencies of over 1010 transformants/µg. The data shown in Figure 3 indicate that approximately 1.5 hours on ice is optimum for M. smegmatis. Increasing the incubation on ice for longer than 1.5 hours leads to a decrease in the transformation efficiency, most likely caused by excessive cell lysis.
We have described a study of some of the factors that influence the electro-transformation efficiency of mycobacterial cells using M. smegmatis as the model organism. Use of the optimal conditions that were elucidated through these experiments should allow one to obtain high transformation efficiencies. The following conditions are recommended: 1) removing cells from their incubator in the log phase of growth; 2) allowing these cells to equilibrate on ice for approximately 1.5 hours before centrifugation; and, 3) electroporating the washed cells at a time constant between 15 to 25 msec (25 µF, 1,000 Ω in parallel). A more detailed protocol that has given high transformation efficiencies in our laboratory is shown in Table 1. Since mycobacterial cells can withstand high voltages as well as long time constants, it is possible that use of capacitance higher than 25 µF or parallel resistance greater than 1,000 Ω may allow further improvement in the transformation efficiencies of mycobacteria.
Table 1. Protocol for high efficiency electro-transformation of M. smegmatis
- Inoculate 1 liter of M-ADC-TW with M. smegmatis.
- Incubate at 37 °C with constant shaking until A600 = 0.2-1.0 (1-2 days).
- Incubate on ice for 1.5 hours.
- Harvest in 250 ml centrifuge bottles by spinning for 10 minutes at 5,000 x g at 4 °C. Discard supernatant. Resuspend each pellet in an equal volume (250 ml) of 10% glycerol. Centrifuge again.
- Resuspend each pellet in 10 ml ice-cold 10% glycerol. Combine cells and transfer to two 50 ml conical polypropylene tubes. Bring the volume in each tube to 50 ml with ice-cold 10% glycerol. Centrifuge for 10 minutes at 2,000 x g at 4 °C.
- Resuspend each pellet in 50 ml ice-cold 10% glycerol. Centrifuge as above for 15 minutes.
- Resuspend each of the two pellets in 10% glycerol for a final volume of 1 ml. At this time the cells may be aliquoted in 1.5 ml polypropylene tubes and frozen quickly in dry ice/ethanol for use at a later date. This protocol may also be continued with the freshly prepared cells.
- Place 50 to 100 µl of cells and DNA (between 5 pg and 5 µg; 5 µl maximum volume) in a 1.5 ml polypropylene tube. Mix well by gently pipetting. Incubate on ice for 1 minute.
- Set the Gene Pulser apparatus to 2.50 kV and 25 µF. Set the Pulse Controller to 1,000 Ω.
- Transfer the cell/DNA mixture to a pre-chilled 0.2 cm electrode gap cuvette. Tap the cuvette on the counter to make sure the cells are at the bottom of the cuvette, and also to remove as many air bubbles as possible.
- Place the cuvette in the white slide and position it at the back of the shocking chamber. Deliver one pulse (time constant 15.0-25.0 msec; field strength 12.5 kV/cm).
- Add 1 ml of M-ADC-TW medium, gently suspend the cells, and transfer to a 15 ml round-bottom polypropylene tube.
- Incubate at 37 °C with constant shaking for approximately 2 hours.
- Plate cells on selective media.
References
- Jacobs, Jr., W. R., Tuckman, M. and Bloom, B. R., Introduction of foreign DNA into mycobacteria using a shuttle phasmid, Nature, 327, 532-535 (1987).
- Snapper, S. B., Melton, R. E., Mustafa, S., Kieser, T. and Jacobs, Jr., W. R., Isolation and characterization of efficient plasmid transformation mutants of Mycobacterium smegmatis, Molec. Microbiol., 4, 1911-1919 (1990).
- Dower, W. J., Miller, J. F. and Ragsdale, C. W., High efficiency transformation of E. coli by high voltage electroporation, Nucl. Acids Res., 16, 6127-6145 (1988).
- Jacobs, Jr., W. R., Kalpana, G. V., Cirillo, J. D., Pascopella, L., Snapper, S. B., Udani, R. A., Jones, W., Barletta, R. G. and Bloom, B. R., Genetic systems for mycobacteria, Meth. Enzymol., 204, 537-555 (1991).
- Stover, C. K., de la Cruz, V. F., Fuerst, T. R., Burlein, J. E., Benson, L. A., Bennett, L. T., Bansal, G. P., Young, J. F., Lee, M. H., Hatfull, G. F., Snapper, S. B., Barletta, R. G., Jacobs, Jr., W. R. and Bloom, B. R., New use of BCG for recombinant vaccines, Nature, 351, 456-460 (1991).
Featured Content
Transfection Solutions

Gene Pulser Xcell Electroporation System
Tried and trusted by thousands of researchers worldwide, transfect every cell type from primary, suspension, and difficult to transfect cells with the Gene Pulser Xcell Electroporation System.

Questions about transfection?

