Why Test for Legionella?

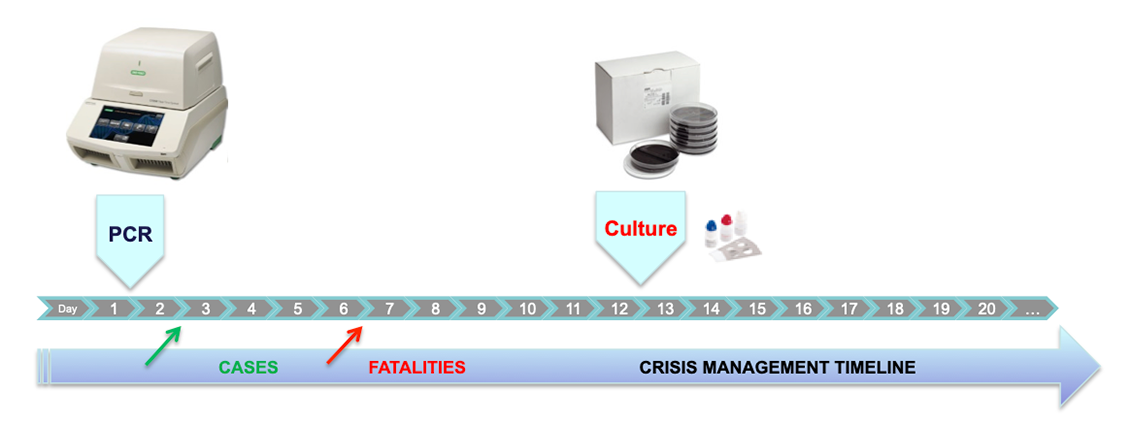
Legionellosis is an infection caused by Legionella bacteria in a water system, such as fountains, misting apparatus and cooling towers, particularly in health establishments, public buildings, and recreational water systems. The bacteria can cause acute pneumonia, Legionnaires' disease and a milder form of pulmonary infection, Pontiac fever. The actual number of Legionnaires' cases is thought to be much higher than reported as many people with pneumonia may not be tested for Legionella infection. Legionella pneumophila is responsible for approximately 90% of the clinical cases, and more than 85% of cases are caused by isolates from serogroup 1. Regular control of the presence of Legionella in water supply systems is an important way of preventing the disease. One of the most sensitive and specific methods developed for Legionella pneumophila testing is PCR (polymerase chain reaction). To ensure accuracy of data, commercially available detection methods must comply with specific requirements and validations recommended by government agencies and professional organizations that have published guidelines not only for the design, but also the commissioning requirements for cooling towers and water systems.
Bio-Rad’s iQ-Check method has been used for over 15 years to monitor Legionella in various water systems including cooling towers, and has been demonstrated to be a very powerful tool for industry and government agencies by safeguarding public health and ensuring environmental safety.
Interested in Talking with a Legionella Expert?
Contact one of our Legionella specialists today to learn more.
Featured Content
iQ-Check Methods

The iQ-Check Legionella solution is an is an ISO/TS 12869 third-party validated method based on amplification and detection of genomic sequences by real-time PCR. The Aquadien Kit, part of the ISO/TS 12869 validation, allows an optimal DNA extraction and purification from bacteria present in all types of water samples for PCR detection after a filtration step. The principle of the extraction is based on alkaline lysis of bacteria in presence of thermal shock and DNA purification using ultrafiltration. The Aquadien W2 Wash Solution is optimized for dirty water samples including those containing potential PCR inhibitors such as additives and chemicals used for water treatment. The iQ-Check Kit contains ready-to-use reagents required to perform the analysis of samples. Additional standards are provided for quantification.
Key benefits:
Quickly screen samples
Results in hours, not days
The only third-party validated solution that include management of free DNA
Protocols adapted to all types of water
Protocol for confirming colonies from standard method agars
Maximum flexibility and automated analysis, including standard curve recall
Featured Content
Free DNA Removal Solution

Water filtration


FDRS treatment


Aquadien protocol
One of the primary challenges when performing PCR for environmental water testing is the potential presence of free DNA, which can lead to an overestimation of the level of targeted DNA sequences. When quantifying Legionella, culture and PCR levels can differ from each other by up to 2 logs. To improve Legionella testing in water, a supplemental step using the Free DNA Removal Solution (FDRS) can be easily integrated in the Aquadien DNA Extraction Kit Workflow. FDRS is a non-carcinogenic safe way to remove free DNA from dead cells in environmental water samples. According to the principle, free DNA is degraded by an enzymatic treatment of the sample prior to DNA extraction, and inactived by the Aquadien Extraction Kit solution, allowing for the extraction of DNA from intact and living cells. The inclusion of FDRS in the iQ-Check workflow reduces the occurrence of false positives. The use of Free DNA Removal Solution is part of the third-party ISO/TS 12869 validation.
XP-Design Assay

L. pneumophila SG1
The XP-Design Assay L. pneumophila SG1 utilizes real-time PCR technology to provide highly accurate identification of Legionella pneumophila serogroup 1 (SG1) in water samples. Following a presumptive positive result from the iQ-Check Legionella Kit ( Legionella spp. and/or L. pneumophila), the XP-Design Assay L. pneumophila SG1 can use the same DNA extract from the Aquadien DNA extraction protocol to deliver L. pneumophila SG1 results within 1 hr. It can also be used directly on isolated colonies to identify and confirm L. pneumophila SG1 present in water samples.
- Results available in 1 hr
- High accuracy and selectivity
- Optimized for use with the iQ-Check Legionella method
- Useful for early detection of L. pneumophila SG1 to improve public health measures, reducing the spread of infection and preventing larger outbreaks
Legionella

- GVPC and BCYE Agars can be used as part of ISO 11731 and NF T90-431
- BCYE Agar is available as individual, with and without L-cysteine
Standard Media
Bio-Rad, as a key partner for water testing, also manufactures a large range of products and solutions for Legionella testing such as GVPC and BCYE medium for the enumeration of Legionella according to ISO 11731.
Contact your Bio-Rad sales specialist for more information












