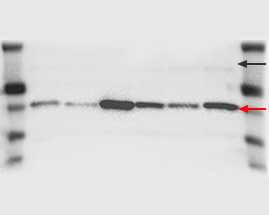
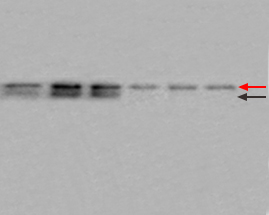
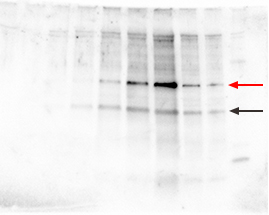
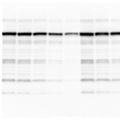
| Multiple nonspecific bands on the blot may be due to antibodies of poor quality or at too high a concentration, insufficient blocking, or nonspecific binding due to the presence of SDS. |
 |
| Possible causes: |
Solutions: |
| Primary antibody concentration too high or cross-reactivity with similar epitopes on other proteins |
|
| Secondary antibody concentration too high, leading to nonspecific binding |
- Decrease or optimize the concentration of the secondary antibody, e.g., using a checkerboard screening protocol
- Use an affinity-purified secondary antibody
- Repeat immunodetection with secondary antibody alone to check for nonspecific binding
|
| Protein exists in several different isoforms |
- Check research literature for existence of isoforms or variants
|
| Primary or secondary antibody contaminated with nonspecific IgG or with IgG cross-reactive among species
|
- Use purified IgG primary antibody fractions and affinity-purified blotting-grade cross-adsorbed secondary antibody
|
| Monoclonal antibodies reacted nonspecifically with SDS-denatured proteins |
- Compare the binding of other monoclonal or polyclonal antibodies
- Blot native proteins as a comparison, e.g., by blue native PAGE
|
| Nonspecific interactions occurring due to ionic associations; for example, avidin, a glycosylated protein, may bind to more acidic proteins on blots |
- Increase washing stringency:
- Increase the ionic strength of the incubation buffers
- Increase the salt concentration of your TBS-T
- Try PBS-T instead of TBS-T (do not do this if using phosphospecific antibodies)
- Increase the duration of washes
- Increase the number of washes
- Perform washes at room temperature
- Include progressively stronger detergents in the washes; for example, SDS is stronger than Nonidet P-40 (NP-40), which is stronger than Tween-20
- Include Tween 20 in the antibody dilution buffers to reduce nonspecific binding
- Increase the Tween-20 concentration to 0.01–0.5% (v/v)
|
| Insufficient blocking of nonspecific sites |
- Increase the concentration of blocking reagent (e.g., BSA, nonfat dry milk, etc.) from 5% to 7% (w/v)
- Consider blocking overnight at 4ºC or at least 1 hour at room temp (increase length of incubations if necessary)
- If not already included, add up to 0.01–0.5% Tween-20 to blocking buffer
- Prepare antibody dilutions to the same blocking buffer with same increased concentration of Tween-20
|
| SDS caused nonspecific antibody binding to immobilized proteins |
- Be sure to equilibrate gel with transfer buffer before transfer
- If the transfer buffer contains SDS, be sure to include a wash step before performing the first antibody incubation step; wash step can be performed with washing buffer
- If washing does not resolve the problem, consider avoiding SDS during blotting procedure if possible
|