The use of reverse transcription (RT) prior to PCR allows for the quantification of RNA targets. Here, reverse transcription is used to make complimentary DNA (cDNA) from RNA and the cDNA target can be amplified and detected through PCR to provide a quantitative measure of the original RNA target.
Bio-Rad’s Droplet Digital™ PCR (ddPCR™) System supports quantitative RNA analysis for gene expression and other applications. Bio-Rad ddPCR Gene Expression Assays are available as probe and EvaGreen assays and protocols are provided for use in one-step or two-step RT-ddPCR protocols. Digital PCR technology can be applied to the detection and quantification of various types of RNA targets (i.e., mRNA, miRNA, siRNA, circulating RNA, viral RNA, etc.) and is used to support the research, manufacturing, and production of RNA therapeutics.
Page Contents
Most analysis of gene expression is focused on quantifying transcription levels. Determination of the presence or absence of particular RNA transcripts and their quantification by reverse-transcription PCR (RT-PCR) is now routine in many laboratories. Digital PCR can increase the detection of rare transcripts and provide easier and more accurate quantitation. Either one-step or two-step RT-PCR can be used in ddPCR.
In Bio-Rad's Droplet Digital™ PCR (ddPCR™) System, each sample is partitioned into many droplets prior to amplification. After cycling, the level of fluorescence is measured in each droplet; droplets with fluorescence are scored as positive, and those with little or no fluorescence are scored as negative. Application of Poisson statistical analysis yields absolute quantification. ddPCR technology does not rely on a standard curve and is insensitive to the number of amplification cycles. One of the greatest advantages of digital PCR for gene expression studies is the exquisite sensitivity of quantification it provides. This sensitivity is particularly evident in quantification of low-abundance target, and in discrimination of rare alleles against an abundant wild-type background.
Bio-Rad ddPCR technology provides absolute quantification of RNAs and gene expression for low copy number templates.
-
RNA sample with target sequence is partitioned into droplets
-
Target and background RNA are randomly distributed among 20,000 droplets
-
Target sequence is amplified by end-point PCR in each droplet
-
Positive droplets are counted to give precise quantification of target sequences in sample
Most analysis of gene expression is focused on quantifying transcription levels. Determination of the presence or absence of particular transcripts and their quantification by reverse-transcription PCR (RT-PCR) is now routine in many laboratories. Digital PCR can increase the detection of rare transcripts and provide easier and more accurate quantitation. Either one-step or two-step RT-PCR can be used in ddPCR.
The discovery of miRNAs added another level of complexity to studies of gene expression. Changes in miRNA levels are thought to be involved in all aspects of gene expression regulation and associated with normal developmental processes as well as diseases ranging from cancer to diabetes. Levels of miRNAs vary over time, and some mature miRNAs display very rapid turnover, necessitating fast, sensitive, and accurate detection.
Recently, miRNAs have been detected in blood. Circulating miRNAs have been shown to be stable and suitable for use as markers of disease including cancer (Mitchell et al. 2008). The detection of miRNA in blood and other fluids such as saliva has paved the way for the development of sensitive new and complementary methods for the diagnosis and monitoring of many diseases. ddPCR has been demonstrated to be an excellent tool for the study of miRNAs (Hindson et al. 2013)
Regulation of gene expression at the genomic level is primarily controlled by the methylation of genomic DNA and by DNA-histone interactions, which are affected by histone modifications such as methylation, phosphorylation, and acetylation. Genomic DNA methylation usually occurs at CpG islands in noncoding regions and generally reduces gene expression.
PCR is one of several techniques used for studying methylation. The most common PCR-based method for the quantification of DNA methylation is the use of different primer pairs that are designed to distinguish methylated from unmethylated CpG islands after treatment with sodium bisulfite (Ku et al. 2011). Unmethylated CpG is converted to UpG by sodium bisulfite, whereas methylated DNA is unaffected. Due to its great capacity to discriminate alleles, digital PCR can provide more sensitive detection and accurate measurement of methylation events. Its extreme precision also makes it a method of choice when operating with low amounts of starting material (such as chromatin immunoprecipitation experiments).
RNA-based biotherapeutics provide a new approach to effect clinically meaningful and previously “undruggable” targets
Drug development has been focused on traditional small molecule and protein therapies for many years, but these approaches have provided means to address only a small fraction of desired targets and diseases. Discovery of the RNA interference (RNAi) process opened a new door and approach to therapeutics discovery and development. The use of RNA as a therapeutic to effect clinically meaningful targets is an exciting new area of work with huge potential as it is positioned to provide a rapid, cost effective, and readily adaptable way to effect previously “undruggable” targets.
Types of RNA therapeutics and mechanism of action
Different types of RNA can be used to activate or inhibit the expression of protein targets in cells to produce a therapeutic effect.
Types of RNA:
- Small activating RNA (saRNA)
- Antisense oligonucleotide (ASO)
- Micro RNA (miRNA)
- Small interfering RNA (siRNA)
- Messenger RNA (mRNA)
RNA therapeutics leverage the RNA interference (RNAi) process that was first discovered in 1998. Since its discovery the scientific community has embraced the opportunity that the RNAi process provides to develop a strong pipeline of RNA-based therapies. The community has worked to overcome challenges (avoiding degradation by RNases, effective delivery to cells, decreasing cytotoxicity, etc.) to develop numerous candidates therapies to address a variety of diseases and disorders.
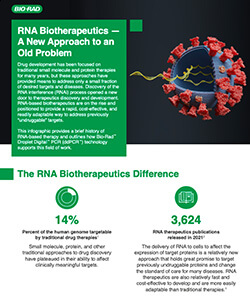
Access infographic about RNA Biotherapeutics – A New Approach to an Old Problem, which includes history and a list of approved RNA therapeutics and those in current trials
-

Download the ebook to read more about the next era in RNA Therapeutics
While many advancements are being made, the field of RNA therapeutics is still new and working to address challenges to make RNA-based therapies safer and more viable on a larger scale.
Droplet Digital PCR technology provides precision and accuracy necessary to address common development, production, and testing challenges and allow RNA therapeutic developers to be confident in their products and maintain workflow efficiency.
Bio-Rad ddPCR technology supports RNA therapeutics across development, production, and testing.
Development
- Quantitative evaluation of RNAs and gene expression to identify potential therapeutic targets
- RNA quantification for analyzing biodistribution
Production
- RNA quantification for dosing
- Ratio analysis testing for multivalent RNA therapeutics
- Poly(A) tail level determination
- Residual host cell DNA quantification and sizing
- Contamination detection (e.g., Mycoplasma)
Testing
Before treatment: Quantitative evaluation of baseline RNAs and gene expression levels
After treatment: Quantitative re-evaluation of RNAs and gene expression levels
-
Featured VideoDid You Know that Droplet Digital PCR Technology is Helping Fuel the RNA Therapeutics Revolution?
-
Featured VideoDid You Know That ddPCR Technology Can Be Used to Characterize the Poly(A) Tail Content in mRNA Products?
Digital PCR can be used for studies of gene expression both alone and in combination with other techniques. One advantage of the QX200™ ddPCR System is that it is compatible with existing primers, probes, and protocols for gene expression studies. It even provides the capacity to do simultaneous detection of target and reference using EvaGreen chemistry (McDermott et al. 2013), making the study of gene expression even simpler and more precise.
References
Hindson CM et al. (2013). Absolute quantification by droplet digital PCR versus analog real-time PCR. Nat Methods 10, 1003–1005. PMID: 23995387
Ku JL et al. (2011). Methylation-specific PCR. Methods Mol Biol 79, 123–32. PMID: 21913069
McDermott GP et al. (2013). Multiplexed target detection using DNA-binding dye chemistry in droplet digital PCR. Anal Chem 8511, 619–11627. PMID: 24180464
Mitchell PS et al. (2008). Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA 105, 10513–10518. PMID: 18663219