Overview
Unity Software Solutions
Unity helps laboratories around the world optimize QC practices and make decisions in real time with tools
to create QC strategies and
manage risk.
Peer Reporting Program
The Unity Interlaboratory Peer Program provides a cost-effective opportunity to compare and improve QC performance by utilizing data generated from thousands of laboratories worldwide.
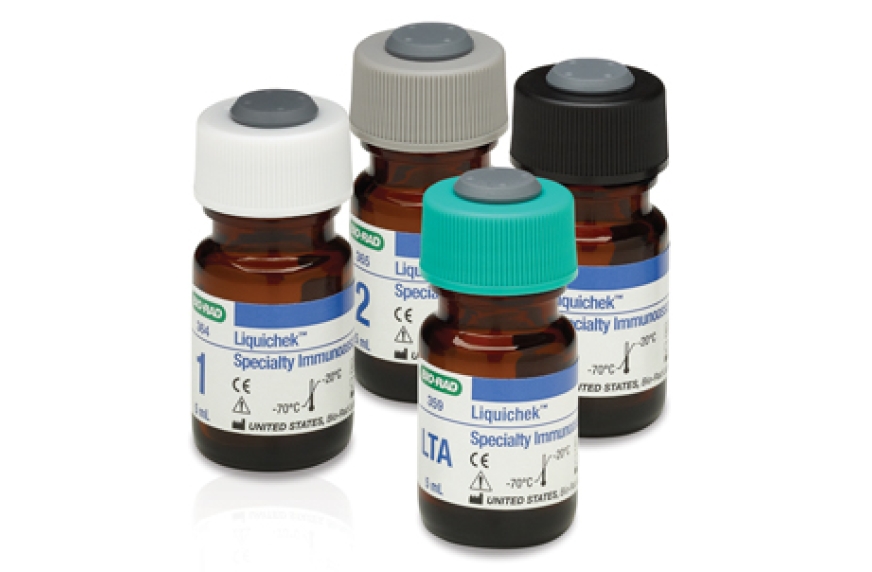
Liquichek Specialty Immunoassay Control is an assayed, liquid, human serum-based control that is designed to complement Lyphochek and Liquichek Immunoassay Plus controls. It includes more complex analytes, such as Intact PTH, Anti-Tg, Anti-TPO, 25-OH Vitamin D, IGF-I, C-Peptide, Osteocalcin and EPO. It offers excellent open-vial stability.
This control covers complex yet high usage specialty immunoassay analytes for laboratories that test specialty analytes for operating rooms, emergency rooms, hospitals and physician offices. The control is available in four levels. Multiple levels of a control allow monitoring of test system reliability.
Order Online—Reserve Your Lots Today
QC Lot reservation and schedule delivery—Now available with online ordering.
Login or Register on My Bio-Rad today and start ordering and reserving »
Features
- Patient-like materials—Liquid, human serum-based control
- Multiple levels—Multilevel control offers monitoring of increased assay range
- Extensive value assignment listings—Allows for use of a wide range of testing methods on most major integrated immunoassay platforms to help consolidate and streamline workflows
- Enables automation—Assayed values available for both automated and manual methods
- Ease of use—Ready-to-use liquid state offers the convenience of not needing to reconstitute
- Long shelf life—Reduces crossover studies which helps simplify workflow
Storage & Stability
| Configuration | Stability | Shelf life | Levels | Format |
|---|---|---|---|---|
| 5 mL | 30 day open-vial stability at 2-8°C for most analytes | 3 year shelf life at -20°C to -70°C | LTA, 1, 2, 3 & Four level MiniPak | Standard vial |
Analytes
-
- 25-Hydroxy Vitamin D
- Anti-Thyroglobulin (Anti-Tg)
- Anti-Thyroperoxidase (Anti-TPO)
- C-Peptide
-
- Erythropoietin (EPO)
- Insulin Growth Factor I (IGF-I)
- Intact PTH
- Osteocalcin
Specialty Immunoassay Controls Analyte Comparison
|
|
|
|
|---|---|---|
| 25-Hydroxy Vitamin D |
|
|
| Anti-Thyroglobulin (Anti-Tg) |
|
|
| Anti-Thyroperoxidase (Anti-TPO) |
|
|
| C-Peptide |
|
|
| Erythropoietin (EPO) |
|
|
| Insulin Growth Factor I (IGF-I) |
|
|
| Intact PTH |
|
|
| Osteocalcin |
|
|
| Procalcitonin |
|
|
| Sex Hormone Binding Globulin (SHBG) |
|
Refer to My eInserts or the product insert (IFU) of currently available lots for specific analyte and stability claims.
Application & Use
This product should be treated the same as patient specimens and run in accordance with the instructions accompanying the instrument, kit or reagent.
Thawed/Unopened
If the product has been stored frozen, allow it to stand at room temperature (18°C to 25°C) for approximately one hour or until completely thawed. Gently swirl the contents until homogeneous with no visible signs of a precipitate. Any precipitate present after thawing should dissolve upon mixing. Store the thawed product at 2-8°C.
Thawed/Opened
If the product has been stored refrigerated, allow it to reach room temperature (18°C to 25°C) before use. Minimize the time at room temperature. Gently swirl the vial several times to ensure homogeneity. After each use, promptly replace the stopper and return to 2-8°C storage.
Aliquot-Frozen
To prepare aliquot samples, allow the control vial to thaw as described above. Gently swirl the vial several times to ensure homogeneity, then dispense the control material into aliquot vials and store at -20°C to -70°C. If sampling aliquot, use it only once. Discard the remaining material.
Related Products
InteliQ Controls
Therapeutic Drug Monitoring Controls
Immunoassay Controls
Frequently Asked Questions
- Room temperature: 18°C to 25°C
- Refrigerator temperature: 2-8°C
- Freezer temperature: -20°C to -70°C/-20°C to -50°C for 2.5 mL vial
Most frozen products recommend avoidance of frost-free freezers. There are exceptions, so refer to specific control insert.
Ordering





Accessories


