Hercules, CA — June 4, 2014 — Bio-Rad Laboratories, Inc. today announced the launch of its SsoAdvanced™ PreAmp Supermix and PrimePCR™ PreAmp Assays (PrimePCR PreAmp Assays are available in the U.S., Canada, Australia, New Zealand, Singapore, and E.U. only). The supermix is formulated to permit unbiased, target-specific preamplification of cDNA or genomic DNA (gDNA), which allows researchers to stretch their limited nucleic acid samples. PrimePCR PreAmp assays are wet-lab validated preamplification primers that work seamlessly with the supermix to ensure optimal preamplification for real-time PCR.
The SsoAdvanced PreAmp Supermix is especially useful for researchers who are working with limited samples or are interrogating multiple targets simultaneously. It enables simultaneous 1,000-fold target-specific preamplification of as little as 100 pg of cDNA (or gDNA) for up to 100 genes while delivering the industry’s lowest bias. It is also more cost-effective than any other preamplification solution available.
The SsoAdvanced PreAmp Supermix can be paired with Bio-Rad’s iScript™ Advanced or iScript Supermix reverse transcription reagents. Using the PreAmp Supermix preamplification protocol (see illustration), researchers first isolate RNA and synthesize cDNA using either of the iScript reagents. After mixing the cDNA (or gDNA) with SsoAdvanced PreAmp Supermix and pooled primers (or with PrimePCR PreAmp assays), PCR is performed to preamplify targets of interest. The reaction is then ready for real-time PCR amplification and detection.
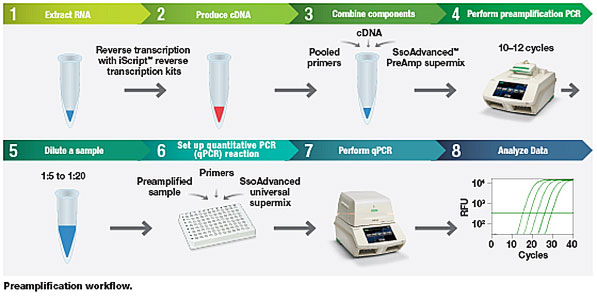
SsoAdvanced PreAmp Supermix preamplification workflow.
For the most accurate gene expression analysis, researchers should pair the SsoAdvanced PreAmp Supermix with PrimePCR PreAmp assays, which are available for every PrimePCR gene expression assay. PrimePCR assays are the only wet-lab validated and specificity-verified primers. The SsoAdvanced PreAmp Supermix is validated for SYBR® Green, probe-based (e.g. TaqMan®), and custom-designed real-time PCR assays.
Visit www.bio-rad.com/PreAmp and www.bio‑rad.com/PrimePCR for more information
About Bio-Rad
Bio-Rad Laboratories, Inc. (NYSE: BIO and BIOb) has been at the center of scientific discovery for 60 years, manufacturing and distributing a broad range of products for life science research and clinical diagnostic markets. The company is renowned for its commitment to quality and customer service among university and research institutions, hospitals, public health and commercial laboratories, as well as the biotechnology, pharmaceutical, and food safety industries. Founded in 1952, Bio-Rad is based in Hercules, California, and serves more than 100,000 research and healthcare industry customers through its global network of operations. The company employs approximately 7,750 people worldwide and had revenues exceeding $2.1 billion in 2013. Visit us at www.bio‑rad.com.
This release contains certain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements generally can be identified by the use of forward-looking terminology such as, “believe,” “expect,” “may,” “will,” “intend,” “estimate,” “continue,” or similar expressions or the negative of those terms or expressions. Such statements involve risks and uncertainties, which could cause actual results to vary materially from those expressed in or indicated by the forward-looking statements. For further information regarding the Company's risks and uncertainties, please refer to the “Risk Factors” in the Company’s public reports filed with the Securities and Exchange Commission, including the Company’s most recent Annual Report on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. The Company cautions you not to place undue reliance on forward-looking statements, which reflect an analysis only and speak only as of the date hereof. Bio-Rad Laboratories, Inc., disclaims any obligation to update these forward-looking statements.
For more information contact:
Sam Ropp
Bio-Rad
510-741-6528
Sam_Ropp@bio-rad.com
Ken Li
Chempetitive Group
312-997-2436 x 112
kli@chempetitive.com
