
Overview
Buy Digital PCR Assays
Simply enter your target of interest or sequence and order a ddPCR Assay
Find an AssayDroplet Digital PCR Publications
Browse the growing list of cited peer-reviewed journal articles in Droplet Digital PCR research featuring Bio-Rad products.
Residual host cell DNA (HCD) that carries over during the process of manufacturing therapeutic proteins and vaccines poses safety concerns and must not exceed levels established by regulatory agencies such as the U.S. Food and Drug Administration and the World Health Organization. Bio-Rad’s ddPCR Residual DNA Quantification Kits are ideal for highly precise quantification of HCD in complex bioprocess intermediates. The kits contain an optimized ddPCR CHO or E. coli Residual Quantification Assay and ddPCR Supermix for Residual DNA Quantification. Both assay and supermix are guaranteed free of contaminating DNA.
Key Benefits
The Residual DNA Quantification Kits are optimized to enable:
- Highly precise, femtogram-level quantification of residual CHO or E. coli DNA
- Direct quantification without DNA purification steps
- Compatible with Bio-Rad’s QX100, QX200, or QX200 AutoDG Droplet Digital PCR Systems
Sample Data
Direct Sample Input: Eliminate DNA Extraction Prior to Residual Host Cell DNA Quantification
Many biopharmaceutical products (biologics, vaccines, etc.) are produced in mammalian or bacterial host cells. DNA levels and matrix conditions vary during the purification process, making residual host cell DNA extraction difficult. Inhibitory substances may also be present, making quantification by qPCR difficult and inaccurate. ddPCR eliminates the need for DNA extraction, as partitioning in ddPCR minimizes the effect of inhibitory substances on the PCR reaction, enabling accurate quantification from complex bioprocess intermediates using direct sample input. Proteinase K and DTT can also be used prior to ddPCR to enable direct input
of samples containing up to 5 mg/ml of protein (Figure 1).
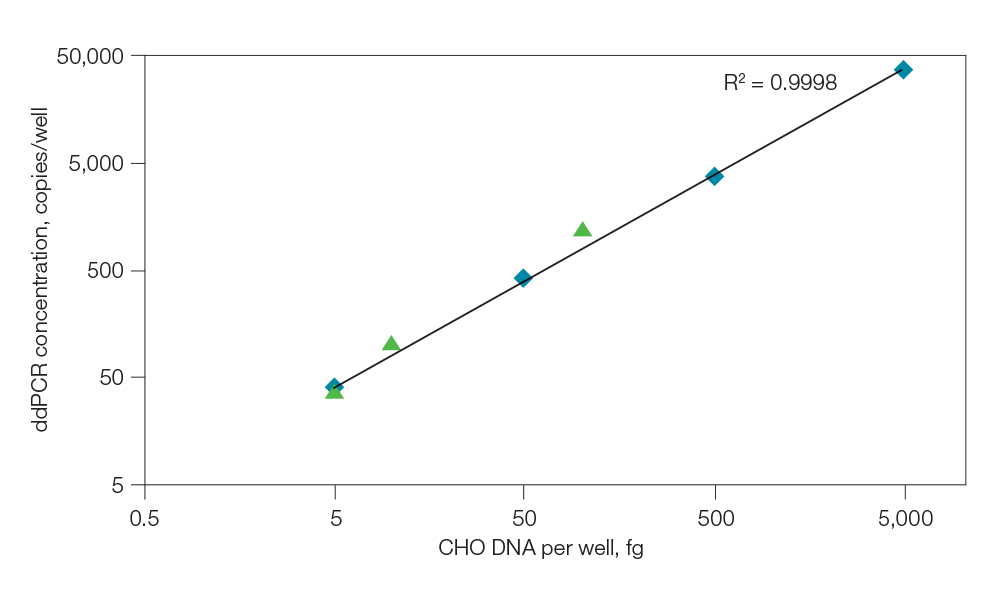
Fig. 1. DNA detection in samples with high IgG levels using Proteinase K pretreatment. CHO DNA was spiked into a sample matrix composed of 100 mg/ml IgG, 3% mannitol, 2% sucrose, 10 mM L-arginine, and 0.01% Tween 20. The sample was then treated with PK and diluted to 100, 10, and 5 fg of CHO DNA prior to ddPCR analysis. CHO standards were also included as controls to determine recovery of the test sample. Full recovery of the CHO DNA was obtained at the three concentrations tested. The data show good linearity, sensitivity, and IgG tolerance. The amount of IgG equivalent in the 100 fg CHO DNA sample is 5 mg/ml. CHO DNA standard ( ); CHO spiked matrix + PK (
); CHO spiked matrix + PK ( ).
).
Sensitive and Precise HCD Quantification
ddPCR CHO and E. coli Residual DNA Quantification Kits enable highly sensitive and precise detection and quantification of host cell DNA without a standard curve. The ddPCR CHO Residual DNA Quantification Kit can reliably detect as little as 1 fg of DNA with a limit of quantification (LOQ) of ≤15 fg per 20 µl reaction and a linear range of 3 fg–3 pg. The ddPCR E. coli Residual DNA Quantification Kit can reliably detect as little as 15 fg of DNA with an LOQ of ≤30 fg per 20 µl reaction and a linear range of 30 fg–3 pg (Figure 2).
Free of CHO and E. coli Contaminating DNA
The ddPCR CHO and E. coli Residual DNA Quantification Assays and ddPCR Residual DNA Quantification Supermixes are guaranteed to be free of contaminating CHO and E. coli DNA.
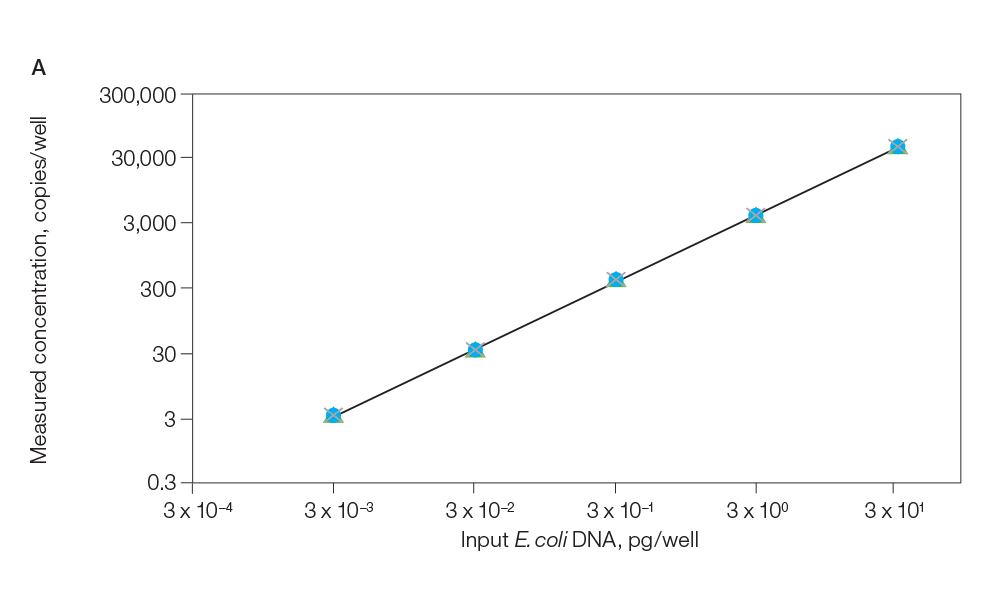
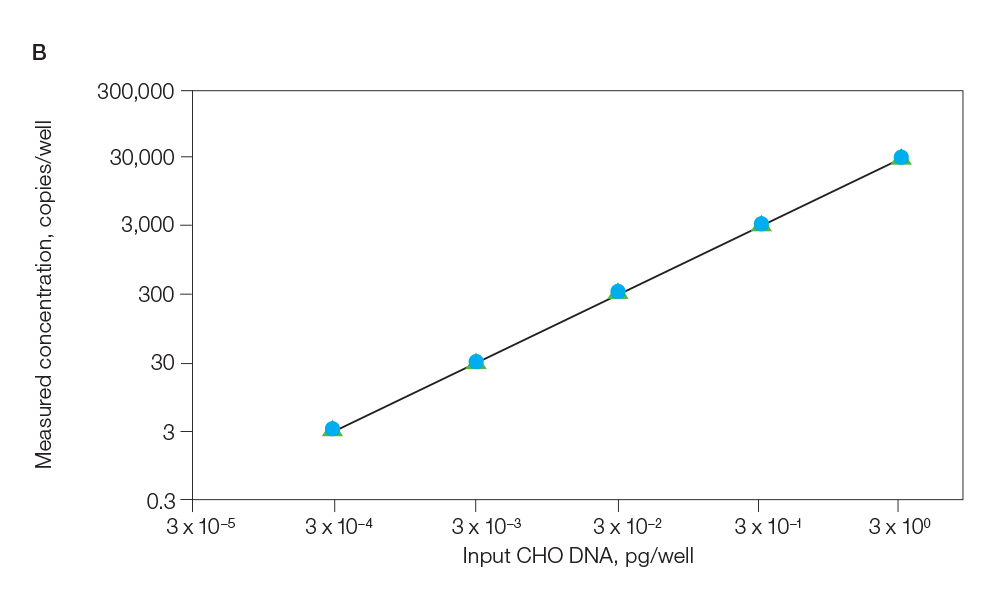
Fig. 2. Titration of E. coli and CHO DNA. A, titration of E. coli DNA from 3 fg to 30 pg; B, titration of CHO DNA from 0.3 fg to 3 pg. Both plots display highly reproducible results between different lots of assays. Lot 1 (![]() ); lot 2 (
); lot 2 (![]() ); lot 3 (
); lot 3 (![]() ).
).
Consistent Performance Even with Highly Fragmented DNA
The ddPCR CHO and E. coli Residual DNA Quantification Assays yield accurate quantification of results, regardless of the size of the DNA molecules. There is less than 20% variation in concentration calls with intact or fragmented DNA.
Resources

Droplet Digital PCR Applications Guide
Read in-depth information about setting up experiments for ddPCR applications.
Rare Mutation Detection Best Practices Guidelines
Learn best practices for performing mutation detection experiments.
Ordering

200 x 20 µl reactions, includes 20x E. coli Residual DNA Quantification Assay and ddPCR Supermix for Residual DNA Quantification. For use with the QX100™, QX200™, or AutoDG™ Droplet Digital™ PCR Systems

200 x 20 µl reactions, includes 20x CHO Residual DNA Quantification Assay and ddPCR Supermix for Residual DNA Quantification. For use with the QX100™, QX200™, or QX200™ AutoDG™ Droplet Digital™ PCR Systems




