Transfection of cells can be accomplished by various methods, including chemical, biological, and instrument-based. This section provides an overview of the different instrument-based transfection methods available, discusses how they work, and describes their pros and cons.
Related Topics: Chemical- and Viral-Based Transfection Methods, Posttransfection Analysis of Cells and Cell Counting Methods.
Page Contents
Transfection can be accomplished using chemical, biological, or physical methods. Common methods include electroporation, the use of virus vectors, lipofection, and biolistics. Many types of genetic material, including plasmid DNA, siRNA, proteins, dyes, and antibodies may be transfected using any of these methods. However, no single method can be applied to all types of cells; transfection efficiencies and cytotoxicity may vary dramatically and depend on the method, cell type being utilized, and types of experiments being performed. Therefore, to obtain high efficiencies, all relevant factors should be considered in planning and selecting the appropriate transfection method.
| Method | Function | Pros | Cons | Cells | Products |
| Electroporation | Nucleic acids or other molecules are introduced into cells by creating transient pores in the plasma membrane using an electric pulse | Nonchemical method that doesn't seem to alter the biological structure or function of the target cells Easy to perform High efficiency Can be applied to a wide range of cell types |
Cell mortality (if using suboptimal conditions) | Eukaryotic cells (primary, stem cells) Prokaryotic cells (bacteria, yeast) Plant protoplasts |
Gene Pulser Xcell™ electroporation system Gene Pulser MXcell™ electroporation system MicroPulser™ electroporator |
| Biolistic particle delivery | Delivery of nucleic acids into cells via high-velocity nucleic acid-coated microparticles | Simple, rapid, versatile technique Targeted intracellular gene delivery Cell type independent Uses small amounts of DNA Delivers single or multiple genes No carrier DNA needed Can deliver large DNA fragments No extraneous genes or proteins delivered Requires little manipulation of cells High reproducibility |
Generally lower efficiency compared to electroporation or viral or lipid mediated transfection Limited bacterial transfection data Requires the preparation of microparticles Instrument cost Requires purchase agreement |
Plant Primary cells Tissue In vivo applications |
Helios® gene gun system PDS-1000/He and Hepta™ systems |
| Microinjection | Direct injection of naked DNA | Can be used for many animals | Laborious (one cell at a time) Technically demanding and costly |
Eukaryotic cells | |
| Laserfection/ optoinjection |
Uses laser light to transiently permeabilize a large number of cells in a very short time | Very efficient Works with many cell types Few cell manipulations needed |
Requires cell to be attached Expensive laser equipment required |
Attached cells |
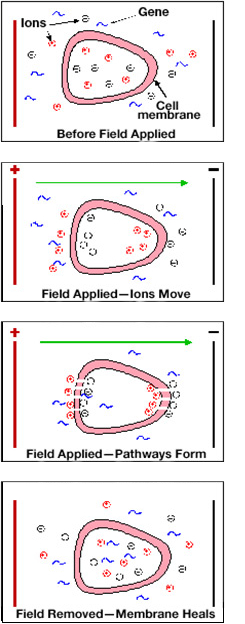
- 1. Electroporation exposes a cell to a high-intensity electric field that temporarily destabilizes the membrane
- 2. During this time the membrane is highly permeable to exogenous molecules present in the surrounding media
- 3. DNA then moves into the cell through these holes
- 4. When the field is turned off, the pores in the membrane reseal, enclosing the DNA inside
Electroporation of cells.
Biolistics is the delivery of nucleic acids into cells by firing nucleic acid-coated microparticles into them.
Helios Gene Gun
- For in situ, in vivo and in vitro transformations
- Applications for animals, plants, cell culture, nematodes, yeast and bacteria
- Pressure range 100–600 psi enables fine-tuning of penetration
- Highly portable can be used in the field
- Small target area for accurate targeting
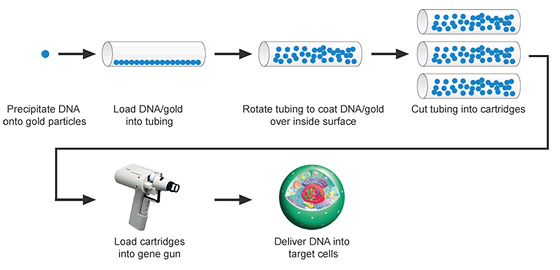
Biolistic particle delivery workflow.
PDS-1000/He Biolistic Particle Delivery System
- For in vitro, ex vivo (and in vivo for some plants and microbes)
- Applications for animal cell and organ culture, plant cell culture and explants, pollen, insects, algae, fungi and bacteria
- Pressure range 450–2200 psi gives flexibility and penetration — ideal for plant applications
- Large target area — more cells can be transformed
Procedure
- DNA-coated microcarriers (thin plastic disk) are spread over the central area of that disk using a pipette tip.
- Disk loaded with the DNA-coated particles is placed into a holder inside the PDS-1000 system.
- The system uses high pressure helium, released by a rupture disk, and a partial vacuum, to propel the macrocarrier sheet loaded with DNA-coated gold macrocarriers toward the target cells.
- Macrocarrier is halted after a short distance by a stopping screen.
- DNA-coated particles continue traveling toward the target to penetrate the cells.
- Sample chamber is subjected to a partial vacuum, from 15 to 29 in. of mercury, depending on the target cells.
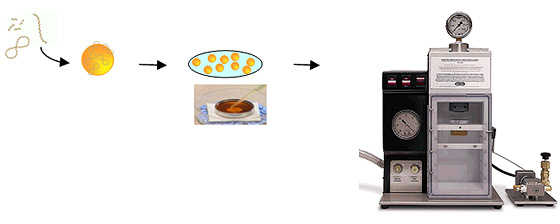
Workflow for delivery using PDS-1000/He system.
- Direct injection of naked DNA
- Laborious (one cell at a time)
- Technically demanding and costly
- Can be used for many animals
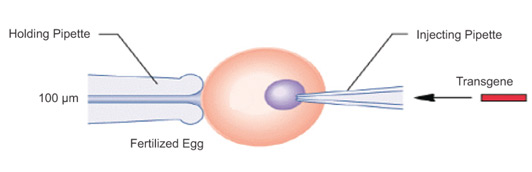
Microinjection of particles.
- This procedure uses laser light to transiently permeabilize a large number of cells in a very short time
- Various substances, including ions, small molecules, dextrans, short interfering RNAs (siRNAs), plasmids, proteins, and semiconductor nanocrystals can be efficiently optoinjected into numerous cell types
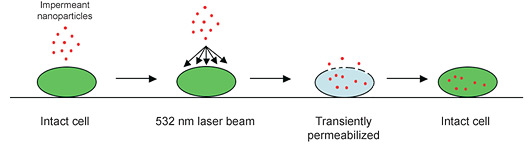
Workflow for laserfection.
The transfection protocol online library contains protocols obtained from the literature, developed by Bio-Rad scientists, or submitted by scientists like you. Browse protocols to view our library and find your starting point or submit a protocol by clicking the proper technology.
Belyansteva IA (2009). Helios Gene Gun-mediated transfection of the inner ear sensory epithelium. Methods Mol Biol 493, 103–123. PMID: 18839344
Benediktsson AM et al. (2005). Ballistic labeling and dynamic imaging of astrocytes in organotypic hippocampal slice cultures. J Neurosci Methods 141, 41–53. PMID: 15585287
Eizema K et al. (2000). Endothelin-1 responsiveness of a 1.4 kb phospholamban promoter fragment in rat cardiomyocytes transfected by the gene gun. J Mol Cell Cardiol 32, 311–321. PMID: 10722806
Fujiki R et al. (2009). GlcNAcylation of a histone methyltransferase in retinoic-acid-induced granulopoiesis. Nature 459, 455–459. PMID: 19377461
Gildea JJ et al. (2009). Caveolin-1 and dopamine-mediated internalization of NaKATPase in human renal proximal tubule cells. Hypertension 54, 1070–1076. PMID: 19752292
Helledie T et al. (2008). A simple and reliable electroporation method for human bone marrow mesenchymal stem cells. Stem Cells Dev 17, 837–848. PMID: 18752428
Hockemeyer D et al. (2009). Efficient targeting of expressed and silent genes in human ESCs and iPSCs using zinc-finger nucleases. Nat Biotechnol 27, 851–857. PMID: 19680244
Huang B et al. (2008). RNA interference-mediated in vivo silencing of fas ligand as a strategy for the enhancement of DNA vaccine potency. Hum Gene Ther 19, 763–773. PMID: 18627219
Shimamura K et al. (2007). Generation of secondary small interfering RNA in cell-autonomous and non-cell autonomous RNA silencing in tobacco. Plant Mol Biol 63, 803–813. PMID: 17225952
Su L et al. (2009). Neural stem cell differentiation is mediated by integrin beta4 in vitro. Int J Biochem Cell Biol 41, 916–924. PMID: 18834954
Tseng CN et al. (2013). A method to identify RNA A-to-I editing targets using I-specific cleavage and exon array analysis. Mol Cell Probes 7, 38–45. PMID: 22960667
Wirth MJ and Wahle P (2003). Biolistic transfection of organotypic cultures of rat visual cortex using a handheld device. J Neurosci Methods 125, 45–54. PMID: 12763229
Zhang G and Selzer ME (2001). In vivo transfection of lamprey brain neurons by gene gun delivery of DNA. Exp Neurol 167, 304–311. PMID: 11161618
Related Content
Videos
This tutorial highlights the main components and features of the Gene Pulser Xcell system. It provides information about system installation and the setup of electroporation experiments, including important troubleshooting tips and answers to frequently asked questions. Ordering information for system components and accessories is also provided.
Documents
TEST
| 6176 | Electroporation Systems Overview | Click to download |
| 6177 | Biolistic Particle Delivery Systems | Click to download |
| 6178 | Recommended Biolistic System by Cell Types | Click to download |
| 6179 | Lipid Transfection Reagents Selection Guide | Click to download |

