Following 2-D electrophoresis, proteins are visualized through in-gel staining. The type of stain used varies depending on the conditions and goals of your experiment and the downstream applications.
Whether your focus is sensitivity or throughput, Bio-Rad has a number of stains available to satisfy any set of experiment requirements. From the popular Bio-Safe™ Coomassie Blue Stain to the more sensitive Oriole™ Gel Stain, Bio-Rad's visualization products ensure proper protein detection and analysis. In addition, Bio-Rad offers accessory products to facilitate handling of large or fragile gels during the staining process, such as the Dodeca™ Stainers, which are designed for reproducible staining of up to 12 gels at a time.
This section provides guidelines for the detection of proteins in gels using different protein stains and stainers from Bio-Rad. It also provides protocols for total protein staining.
Related Topics: Imaging and Analysis of 2-D Electrophoresis Gels, Protein Spot Excision and Protein Identification, Troubleshooting 2-D Electrophoresis Gels with 2-D Doctor™, Protein Sample Preparation for 2-D Electrophoresis, First Dimension Separation (Isoelectric Focusing), and Second-Dimension Separation.
Page Contents
Gels are run for either analytical or preparative purposes. The intended use of the gel determines the amount of protein to load and the means of detection. It is most common to make proteins in gels visible by staining them with dyes or metals. Each type of protein stain has its own characteristics and limitations with regard to the sensitivity of detection and the types of proteins that stain best. Sometimes proteins are transferred to membranes by western blotting to be detected by immunoblotting or total protein stain. Western blotting of 2-D gels is often used for analysis of posttranslational modificatons.
If the purpose of gel electrophoresis is to identify low-abundance proteins (low-expression protein in a cell extract or contaminants in a purification scheme), then a high protein load (0.1–1 mg/ml) and a high-sensitivity stain, such as silver or a fluorescent stain, should be used. When the intention is to obtain enough protein for use as an antigen or for sequence analysis, then a high protein load should be applied to the gel and the proteins visualized with a staining procedure that does not fix proteins in the gel. Quantitative comparisons require the use of stains with broad linear ranges of detection.
The sensitivity that is achievable in staining is determined by:
- the amount of stain that binds to the proteins
- the intensity of the coloration
- the difference in coloration between stained proteins and the residual background in the body of the gel (the signal-to-noise ratio)
Unbound stain molecules can be washed out of the gels without removing much stain from the proteins.
Proteins vary in their interactions with stains. A protein spot that can be visualized using one staining protocol may not be visible when a different stain is applied. Therefore, it is important to evaluate the efficacy of multiple stains with each new sample and select the stains that generate the best results. All stains interact differently with different proteins. No stain will universally stain all proteins in a gel in proportion to their mass. The only observation that seems to hold for most stains is that they interact best with basic amino acids. For critical analysis, replicate gels should be stained with two or more different stains.
Of all available stains, colloidal Coomassie Blue (QC Colloidal Coomassie) appears to stain the broadest spectrum of proteins. It is instructive, especially with 2-D PAGE gels, to stain a colloidal Coomassie Blue-stained gel with silver or to stain a fluorescently stained gel with colloidal Coomassie Blue or silver. Very often, this double staining procedure will show a few differences between the protein patterns. It is most common to stain gels first with Coomassie Blue or a fluorescent stain, and restain with silver. However, the order in which the stains are used does not seem to be important, as long as the gels are washed well with high-purity water between stains.
Silver stains are very sensitive; however, they have a limited dynamic range and do not react with all proteins uniformly. The more sensitive Flamingo™ Fluorescent Gel Stain is ideal for nonspecific visualization and quantitation of proteins in SDS-PAGE gels. It uses an easy two-step protocol that can be completed in as little as 5 hours and does not require destaining. The steps are not time-sensitive and the dye will not overstain the gels. Flamingo is suited for use with high-performance laser-based imaging systems. Oriole Fluorescent Gel Stain is an easy-to-use, fast, sensitive, and highly linear protein gel stain for visualization and quantitation of proteins in SDS-PAGE gels. The simple protocol requires a one-step staining process without prior fixing. Since destaining is not required, protein samples can be accurately visualized and quantitated in less than 2 hours. The excitation and emission characteristics of Oriole Stain make it ideal for UV-based imagers like ChemiDoc™ Imaging Systems and Gel Doc™ gel documentation systems.
Bio-Rad's proprietary stain-free imaging technology enables visualization of protein gels and bots without a staining step. Bio-Rad offers mini- and midi-format precast gels and acrylamide solutions containing a unique compound that provides stain-free protein detection. There is no need for a destaining step, and visualization can be done within 5 minutes. The sensitivity of stain-free imaging is comparable to that of Coomassie.
Bio-Rad's stain-free enabled imagers (ChemiDoc™ MP, ChemiDoc™, ChemiDoc™ XRS+, Gel Doc™ XR+, and Gel Doc™ EZ Systems) come with automated imaging and analysis software and give less background compared to traditional Coomassie staining procedures, thereby reducing variability between and within gel blots.
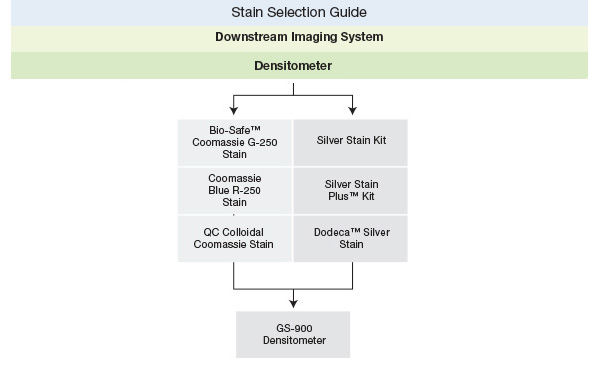
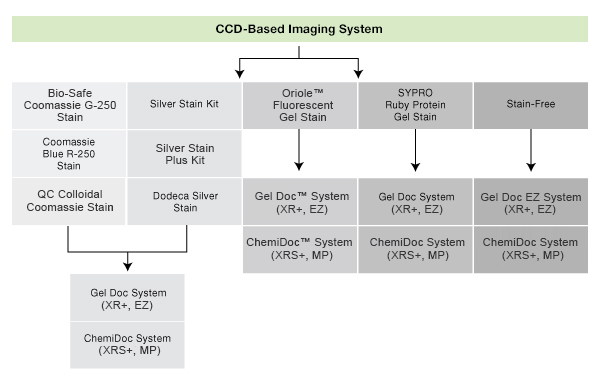
Visit the Protein Staining page to learn more about the different types of protein stains for SDS-PAGE gels pictured below and choose the best stain for your application, and see the Protein Blotting Detection and Imaging page for information about staining and other methods to detect proteins on western blots.


Dodeca Stainers
Staining can generally be performed in a tray, but when staining a large number of gels, it is most efficient to use gel stainers such as the Dodeca™ Stainers, which can accommodate up to 12 large-format gels simultaneously. The Dodeca Stainers are compatible with Bio-Safe Coomassie Stain, Coomassie Blue R-250 stain, SYPRO Ruby Protein Gel Stain, Oriole Fluorescent Gel Stain, Flamingo Fluorescent Gel Stain, and the Dodeca Silver Stain Kit.
Stainer Compatibility with Different Gel Sizes
| Gel Size | (W x L) Gel Format | |
| Large Dodeca Stainer | 25 x 20.5 cm | PROTEAN® Plus handcast (requires one attachment per tray) |
| Small Dodeca Stainer | 20 x 20.5 cm | PROTEAN Plus handcast |
| 18.5 x 20 cm | PROTEAN II XL handcast | |
| 13.3 x 8.7 cm | Criterion (up to 24 gels, require one attachment per tray) |
Documents
TEST
| Number | Description | Options |
|---|---|---|
| 6209 | Total Protein Staining | Click to download |
