- Introduction to Protein Electrophoresis
- Performing Protein Electrophoresis
This section provides tips for performing electrophoresis, loading samples, and troubleshooting, as well as a general protocol for SDS-PAGE.
Related Topics: Protein Electrophoresis Methods, Protein Electrophoresis Equipment, Protein Electrophoresis Sample Preparation, Protein Electrophoresis Reagent Selection and Preparation, and Protein Detection and Analysis.
Page Contents
General Protocol: SDS-PAGE
The following is a generalized protocol for running a Mini-PROTEAN TGX gel in the Mini-PROTEAN Tetra cell.
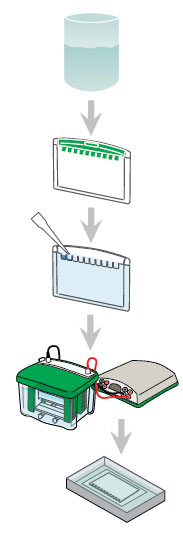 |
Prepare buffers. a. Running buffer (1x): Dilute 100 ml of 10x stock with 900 ml diH2O. b. Sample buffer: Use Laemmli sample buffer. |
|||||||||||||||||||||
| Prepare gels and assemble the electrophoresis cell. a. Remove the comb and tape from the gels and assemble the electrophoresis cell. b. Fill the inner and outer buffer chambers with running buffer. Fill the inner (upper) buffer chamber of each core with 200 ml of 1x running buffer. Fill the outer (lower) buffer chamber to the indicator mark for 2 gels (550 ml) or 4 gels (800 ml) with 1x of running buffer. At runs >200 V, fill the outer buffer chamber to the 4-gel (800 ml) mark.
|
||||||||||||||||||||||
Prepare samples as indicated in the table below.
|
||||||||||||||||||||||
| Heat samples at 90–100°C for 5 min (or at 70°C for 10 min). Load the appropriate volume of your protein sample on the gel. | ||||||||||||||||||||||
| Connect the electrophoresis cell to the power supply and perform electrophoresis according to the following conditions:
|
||||||||||||||||||||||
| After electrophoresis is complete, turn the power supply off and disconnect the electrical leads. Pop open the gel cassettes and remove the gel by floating it off the plate into water. | ||||||||||||||||||||||
| Stain and image the gel, using one of the protocols. | ||||||||||||||||||||||
General Tips for Protein Electrophoresis
- When preparing running buffers, make the solution as written and do not titrate to a pH. The ion balance is set by the concentration of reagents; adjusting the pH alters this balance and leads to undesirable results
- Do not reuse running buffers
- Use 5–10 V per cm gel length for about 10 min during sample entry (or until the sample has concentrated at the starting point of the separation gel). Then continue with the voltage setting recommended in the instruction manual for the electrophoresis system you are using
- Use the voltage setting recommended in the instruction manual for the electrophoresis system you are using; excessive voltage leads to decreased band resolution, band smiling, and lane distortions
- When running multiple cells, use the same voltage for multiple cells as you would for one cell. Be aware that the current drawn will double with two cells (compared to one). Use a power supply that can accommodate this additional current and set the current limit high enough to permit this additional function
- To maximize reproducibility, maintain the temperature of the electrophoresis buffer at ~20°C with the help of a recirculating cooler
General Tips for Sample Loading
- The total protein amount loaded per lane depends on sample complexity and the sensitivity of the staining technique. Using 15–20 µg protein per lane for mini- or midi-format gels is a good starting point for complex protein samples when staining with Bio-Safe™ Coomassie stain. Determine the optimum protein load by running a dilution series of the sample
- For best resolution, load a concentrated sample rather than a diluted amount
- Centrifuge the sample solution for 10–15 min at >12,000 x g at 20°C before loading to remove insoluble material that may clog the pores of the acrylamide gel
- To avoid edge effects, add 1x sample buffer to the unused wells and never overfill wells
- Load samples either before or after placing the electrophoresis modules into the tank. Both methods produce acceptable results. In both cases, fill both the assembly (inner chamber) and the tank (outer chamber) with buffer
- Add running buffer to the cathode buffer reservoir first and then apply the sample on the stacking gel under the electrode buffer. Sample buffer must contain glycerol to stabilize the sample application zone in the sample well of the gel
- Use pipet tips designed for protein sample loading for best results. For example, Bio-Rad's Prot/Elec tips fit easily between vertical slab gel plates of 0.75 mm while maintaining a large bore for fast flow of sample
- Load the samples into the wells with a Hamilton syringe or a pipet with gel loading tips
- Load samples slowly to allow them to settle evenly on the bottom of the well. Be careful not to puncture the bottom of the well with the syringe needle or pipet tip
- If using Bio-Rad's patented sample loading guide, place it between the two gels in the electrode assembly. Sample loading guides are available for 9, 10, 12, and 15-well formats. Use the sample loading guide to locate the sample wells. Insert the Hamilton syringe or pipet tip into the slots of the guide and fill the corresponding wells
Troubleshooting
| Problem | Cause | Solution |
| Current zero or less than expected, and samples do not migrate into gel | Tape at the bottom of precast gel cassette not removed | Remove tape |
| Insufficient buffer in inner buffer chamber | Fill buffer chamber with running buffer | |
| Insufficient buffer in outer buffer chamber | Fill inner and outer buffer chambers to ensure wells are completely covered | |
| Electrical disconnection | Check electrodes and connections | |
| Gels run faster than expected | Running buffer too concentrated and gel temperature too high; incorrect running buffer type used | Check buffer composition |
| Running or reservoir buffer too dilute | Check buffer protocol and concentrate if necessary | |
| Voltage too high | Decrease voltage by 25–50% | |
| Gels run slower than expected | Running buffer too concentrated | Check buffer protocol and dilute if necessary |
| Excessive salt in sample | Desalt sample | |
| Leaking from inner chamber buffer | Incomplete gasket seal | Wet gasket with running buffer before use |
| Improper assembly of gel into the electrode/companion assembly | Ensure that top edge of short plate fits under notch at top of gasket Ensure that top of short plate touches the green gasket |
|
| Leaking from inner buffer chamber | Inner buffer chamber overfilled | Keep buffer level below top of spacer plate |
| Improper assembly | Ensure that U-shaped electrode core gasket is clean, free of cuts, and lubricated with buffer Ensure that short plate is under notch on gasket |
Evaluation of Separation
| Problem | Cause | Solution |
| Diffuse or broad bands | Poor quality acrylamide or bis-acrylamide, incomplete polymerization | Use electrophoresis-grade reagents Check polymerization conditions |
| Old SDS or sample buffer | Prepare fresh solutions | |
| Gel temperature too high | Use external cooling during run or run more slowly | |
| Bands “smile” across gel, band pattern curves upward at both sides of gel | Excess heating of gel; center of gel runs hotter than either end | Check buffer composition; buffer not mixed well or buffer in inner chamber too concentrated Prepare new buffer, ensuring thorough mixing, especially when diluting 5x or 10x stock |
| Power conditions excessive | Do not exceed recommended running conditions. Decrease power setting from 200 V to 150 V or fill outer chamber to within 1 cm of top of short plate | |
| Insufficient buffer | Fill inner and outer buffer chambers to ensure that wells are completely covered | |
| Smiling or frowning bands within gel lane | Overloaded proteins | Load less protein |
| Sample preparation/buffer issues | Minimize salts, detergents, and solvents in sample preparation and sample buffers | |
| Incorrect running conditions | Use correct voltage | |
| Skewed or distorted bands, lateral band spreading | Excess salt in samples | Remove salts from sample by dialysis or desalting column prior to sample preparation |
| Ionic strength of sample lower than that of gel | Use same sample buffer in samples as in gel | |
| Insufficient sample buffer or wrong formulation | Check buffer composition and dilution instructions | |
| Diffusion prior to turning on current | Minimize time between sample application and power startup | |
| Diffusion during migration through stacking gel | Increase %T of stacking gel to 4.5–5%T Increase current by 25% during stacking |
|
| Uneven gel interface | Decrease polymerization rate Overlay gels carefully Rinse wells after removing comb to residual acrylamide |
Videos
Setting Up and Running the Mini-PROTEAN® TGX™ Precast Gels in the Mini-PROTEAN Tetra Cell
This tutorial shows how to use Bio-Rad's Mini-PROTEAN TGX precast gels with the Mini-PROTEAN Tetra cell. Mini-PROTEAN TGX precast gels are long shelf life precast gels that use standard Tris/Glycine buffers.
This tutorial shows how to use Bio-Rad's Mini-PROTEAN TGX precast gels with the Mini-PROTEAN Tetra cell. Mini-PROTEAN TGX precast gels are long shelf life precast gels that use standard Tris/Glycine buffers.
Documents
Number Description Options
6040
Electrophoresis Guide, Interactive PDF, Rev C
1658100
Mini-PROTEAN Precast Gels Instruction Manual and Application Guide
2317
Ready-to-Run Buffers and Solutions Brochure, Rev F
4006028
Instruction Manual, Laemmli Sample Buffer, Rev J
TEST
Number Description Options
| 6199 | Buffer Formulations | Click to download |
| 6202 | General Protocol: SDS-PAGE | Click to download |


