Proteins in a sample can be quantified using different assays. This section describes available assays for protein concentration determination including the classic Bradford and Lowry assays.
Related Topics: Cell Disruption, Protein Solubilization, Removal of Interfering Substances.
Page Contents
Determine the concentration of protein in a sample by protein assay to:
- Ensure that the amount of protein to be analyzed is appropriate for the separation and visualization method
- Facilitate comparison among similar samples; image-based analysis is simplified when equivalent quantities of proteins have been loaded in the lanes of the gel (Berkelman 2008)
The most commonly used protein assays are colorimetric assays in which the presence of a protein stain causes a color change, proportional to the amount of protein in the sample, that can be measured with a spectrophotometer. All protein assays utilize a dilution series of a known protein (usually bovine serum albumin (BSA) or bovine γ-globulin), to create a standard curve from which the concentration of the sample is derived (see Protocol tab). For additional information on protein quantitation assays see Sapan et al. 1999 and Noble and Bailey 2009.
The chemical components of the sample buffer and the amount of protein available for assay dictate the type of assay that may be used (see table and Selection Guide below).
- Bradford assays (Bradford 1976) — this assay is based on an absorbance shift of the Coomassie Brilliant Blue G-250 dye under acidic conditions, when a redder form of the dye is converted into a bluer form upon binding to protein. The increase of absorbance at 595 nm is proportional to the amount of bound dye, and thus to the amount (concentration) of protein present in the sample. In comparison to other protein assays, the Bradford protein assay is less susceptible to interference by various chemicals that may be present in protein samples*
- Lowry (Lowry et al. 1951) — combines the reactions of cupric ions with the peptide bonds under alkaline conditions with the oxidation of aromatic protein residues. The Lowry method is based on the reaction of Cu+, produced by the peptide-mediated reduction of Cu2+, with Folin-Ciocalteu reagent (a mixture of phosphotungstic acid and phosphomolybdic acid in the Folin-Ciocalteu reaction)
- BCA (Bicinchoninic acid, Smith et al. 1985) — BCA reacts directly with Cu+ (generated by peptide-mediated reduction of Cu2+) to produce a purple end product. The reagent is fairly stable under alkaline conditions and can be included in the copper solution to allow a one-step procedure
*The Bradford assay is, however, highly sensitive to ionic detergents like SDS (see table below).
To measure protein concentration in Laemmli buffers, use the RC DC protein assay, which is compatible with reducing agents and detergents. For more information on protein quantitation using colorimetric assays, refer to Bio-Rad bulletin 1069.
Bio-Rad offers a number of protein assays based on the classic Bradford and Lowry assays. The choice of the assay will depend on a number of factors including the concentration of the proteins in the sample and the presence of reducing agents and detergents.
| Characteristics of Bio-Rad's Protein Assays | ||||
| Protein Assay | ||||
| Quick Start™ Bradford | Bio-Rad | DC™ | RC DC™ | |
| Adapted from method of | Bradford (1976) | Bradford (1976) | Lowry et al. (1951) | Lowry et al. (1951) |
| Standard concentration assay | ||||
| Sample volume | 100 µl | 100 µl | 100 µl | 100 µl |
| Linear range | 0.125–1.5 mg/ml | 0.2–1.5 mg/ml | 0.125–1.5 mg/ml | 0.125–1.5 mg/ml |
| Low-concentration assay | ||||
| Sample volume | 1 ml | 800 µl | 200 µl | 200 µl |
| Linear range | 1.25–25 µg/ml | 1.25–25 µg/ml | 5–250 µg/ml | 5–250 µg/ml |
| Microplate assay sample volume | 5 µl | 10 µl | 5 µl | * |
| Minimum incubation time | 5 min | 5 min | 15 min | 15 min |
| Assay wavelength | 595 nm | 595 nm | 650–750 nm | 650–750 nm |
All assays are easy to use, require little reagent preparation, and give accurate and reproducible results.
* To adapt the RC DC assay to a microplate format, follow the micro test tube (microfuge tube) assay protocol in the RC DC instruction manual up to the centrifugation step where the supernatant is discarded. The pellet can then be transferred to the microplate, and the microplate assay protocol in the DC protein assay manual can be followed.
In a typical protein assay, a chemical reagent is added to a protein sample, producing a visible result, such as a color change in the sample solution. This color change is quantitated with a spectrophotometer or microplate reader, and compared to a standard curve of known concentrations of protein versus their absorbance after reaction with the reagent. The amount of protein in the unknown sample is determined by interpolation, reading the concentration of protein on the standard curve that corresponds to its absorbance.
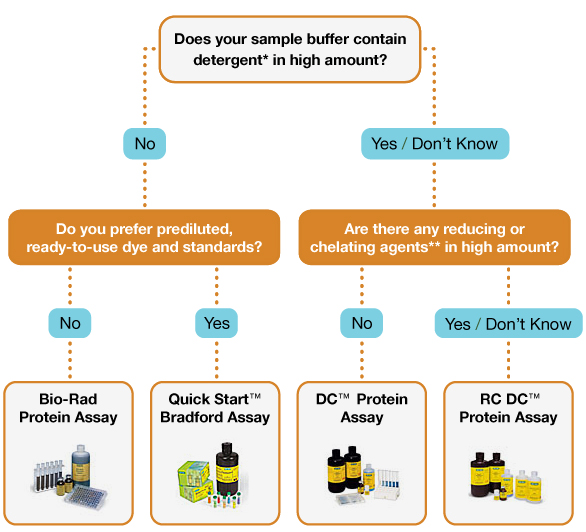
* Nonidet P–40 >0.25%, Triton C-100 >0.05%, Tween 20 >0.01%, SDS >0.025%
** DTT >1 mM, EDTA >1 mM
The color change observed in protein assays is monitored and quantitated using a spectrophotometer. Spectrophotometers such as Bio-Rad's SmartSpec™ Plus Spectrophotometer may have preprogrammed methods for protein quantitation to facilitate data collection and analysis.

SmartSpec Plus spectrophotometer.
Berkelman T (2008). Quantitation of protein in samples prepared for 2-D electrophoresis. Methods Mol Biol 424, 43–49.
Bradford MM (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72, 248–254.
Noble JE and Bailey MJ (2009). Quantitation of protein. Methods Enzymol 463, 73–95.
Sapan CV et al. (1999). Colorimetric protein assay techniques. Biotechnol Appl Biochem 29, 99–108.
Smith PK et al. (1985). Measurement of protein using bicinchoninic acid. Anal Biochem 150, 76–85.
Documents
TEST
| Number | Description | Options |
|---|---|---|
| 6221 | Sample Quantitation (RC DC™ Protein Assay) | Click to download |
